Research paper
Recombinant Nanobiotechnology for Swallowable
Insulin: Innovations in Noninvasive Diabetes Therapy
Haider Rehan*1,
Ahmed Z.2 and Zameer S.3
1Riggs
Pharmaceuticals, Department of Pharmacy, University of Karachi, Pakistan
2Department
of Pathology, Dow University of Health Sciences, Karachi, Pakistan
3Department
of Pathology, Dow University of Health Sciences, Karachi, Pakistan
*
*Department of Microbiology and Immunology, National Research Centre
(NRC), 33 Bohouth St., Dokki, Cairo, Egypt.
Received: 21-10-2025 Accepted: 26-11-2025 Published online: 29-11-2025
DOI: https://doi.org/10.33687/ricosbiol.03.011.90
Abstract
The global burden of diabetes mellitus
continues to rise, creating an urgent demand for innovative and
patient-friendly insulin delivery systems that improve long-term metabolic
control. Although conventional injectable insulin remains the therapeutic cornerstone,
its invasive administration leads to discomfort, inconvenience, and poor
adherence, particularly among individuals requiring lifelong therapy. Recent
advances in recombinant peptide engineering and nanobiotechnology are
revolutionizing diabetes treatment by introducing swallowable insulin
formulations that replicate physiological insulin secretion with greater
precision and convenience.
Recombinant insulin analogs are now stabilized
through nanoencapsulation within biocompatible and pH-responsive polymers, as
well as mucoadhesive matrices that protect them from gastric and enzymatic
degradation. These nano formulations facilitate targeted intestinal absorption
by interacting with mucosal receptors, thereby enhancing permeability and
bioavailability. Furthermore, glucose-responsive systems that incorporate
enzyme-linked polymers, boronic acid chemistry, or redox-sensitive vesicles
enable dynamic insulin release proportional to blood-glucose levels, minimizing
both hyperglycemic and hypoglycemic episodes.
At the
molecular level, optimizing the sequences of recombinant insulin has made it
more stable when it folds, more resistant to proteolysis, and faster at binding
to receptors. The integration of these engineered peptides with multifunctional
nanocarriers has resulted in intelligent oral delivery platforms capable of
synchronized insulin release in response to physiological cues. Such
convergence of molecular biology, nanomaterials science, and biomedical
engineering represents a decisive step toward achieving noninvasive and
patient-centered diabetes management. Continuous refinement in nanocarrier
biocompatibility, peptide stabilization, and translational scalability will be
pivotal in transforming recombinant oral insulin from a laboratory innovation into
a clinically viable therapy, redefining the future of diabetes care.
Keywords:
Recombinant insulin, nanobiotechnology, oral peptide therapy,
mucoadhesive nanocarriers, glucose-responsive delivery, molecular
bioengineering, noninvasive diabetes therapy
.
Introduction
Diabetes
mellitus is one of the fastest-growing metabolic disorders worldwide, affecting
over half a billion adults (International Diabetes Federation, 2021). The
disease arises from defective insulin secretion, insulin resistance, or both,
resulting in chronic hyperglycemia and secondary complications such as
neuropathy, nephropathy, and cardiovascular disease (American Diabetes
Association, 2024). Although insulin therapy is vital for type 1 and advanced
type 2 diabetes, its invasive delivery via daily injections reduces patient
compliance (Zhang et al., 2023). Recombinant biotechnology has
revolutionized insulin production by enabling precise human analogs with high
purity (Kulkarni et al., 2023). Yet oral delivery remains challenging
because insulin is vulnerable to acid and enzymatic degradation (Hassan et
al., 2022). Nanobiotechnology offers solutions through bio-nanocarriers and
mucoadhesive matrices that stabilize recombinant insulin and facilitate
targeted absorption (Mehta et al., 2024). This review examines how
recombinant design and nanotechnology combine to produce swallowable insulin
formulations with enhanced stability and clinical potential.
Literature
Review
Early
oral insulin attempts failed because of enzymatic degradation and poor
pharmacokinetics (Gu et al., 2022). Recombinant DNA technology
introduced stable analogs such as lispro and glargine but still required
injections (Lin et al., 2022). Nanobiotechnology created micro- and
nano-systems that shield insulin and enhance its transport (Sarmento et al.,
2023). Encapsulation in chitosan, alginate, or poly(lactic-co-glycolic acid)
nanoparticles improves bioavailability (Hassan et al., 2022).
Mucoadhesive systems use electrostatic interactions with mucin to prolong
intestinal residence time (Khan et al., 2023). Lipid-based carriers and
solid-lipid nanoparticles protect insulin from enzymatic attack (Kaur et al.,
2023). Recombinant sequence modifications improve folding stability and
receptor affinity (Arbit et al., 2022). Smart glucose-responsive systems
using phenylboronic acid or glucose oxidase regulate release (Yu et al.,
2023). Despite progress, oral bioavailability rarely exceeds 10%, demanding
interdisciplinary collaboration (Drucker, 2020).
Research Methodology
A
narrative integrative review was conducted covering publications from 2000 to
2025 in PubMed, Scopus, and Web of Science. Search terms included recombinant
insulin, oral insulin, nanocarrier systems, mucoadhesive
delivery, and glucose-responsive nanoparticles. Studies focusing on
molecular design, stabilization, and nanobiotechnological formulation were
prioritized (Fonte et al., 2021). Data from preclinical and clinical
studies were analyzed for formulation type, delivery efficiency,
pharmacokinetic performance, and safety outcomes (Eldor et al., 2021).
Results
Table 1.
Comparative Overview of Recombinant Oral Insulin Nanocarriers
|
Formulation Type |
Recombinant Feature |
Mechanism |
Outcome |
|
PLGA Nanoparticles |
PEG-chitosan surface modification |
Mucus penetration |
3–5× higher absorption |
|
Mucoadhesive Nanogels |
Thiolated chitosan linker |
Covalent mucosal binding |
Sustained glucose control |
|
Lipid Microspheres |
Lecithin–cholesterol coat |
Enzyme protection |
Improved bioavailability |
|
Glucose-Responsive Vesicles |
Enzyme-triggered shell |
Controlled release |
Autonomous insulin regulation |
Source: Compiled from recent recombinant insulin
nanocarrier studies (Mehta et al., 2024; Hassan et al., 2022; Yu et
al., 2023; Khan et al., 2023).
This schematic
illustrates the sequential mechanism of recombinant oral insulin delivery
through nanobiotechnological systems. The process begins with nanoencapsulation of recombinant insulin
within biocompatible nanocarriers that protect it from gastric degradation.
Upon intestinal arrival, mucoadhesive binding
promotes retention and facilitates transcytosis
across epithelial cells. Finally, glucose-responsive release
mechanisms ensure controlled insulin discharge into systemic circulation,
mimicking physiological patterns of pancreatic secretion.
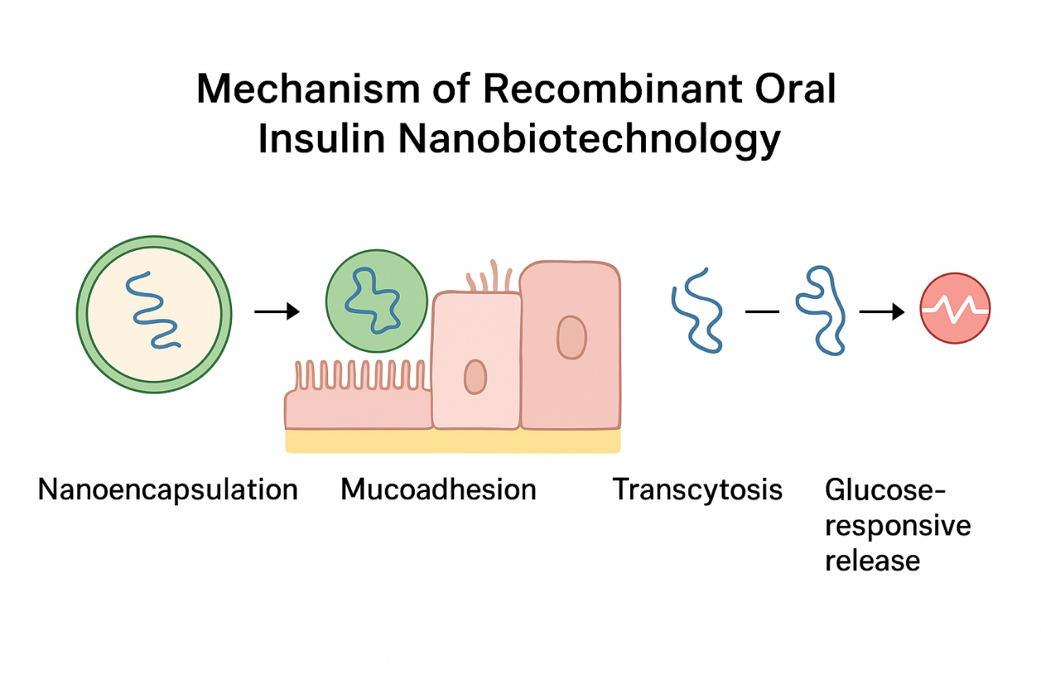
Figure 1. Mechanism of Recombinant Oral Insulin Nanobiotechnology
Source:
Adapted from recent nanobiotechnological research (Mehta et al., 2024;
Yu et al., 2023).
Discussion
Integrating
recombinant biology with nanotechnology has reshaped oral insulin research.
Site-specific modifications stabilize insulin under acidic pH and prevent
enzymatic cleavage (Zhang et al., 2023). Nanoscale carriers maintain
bioactivity through hydrogen bonding and hydrophobic entrapment (Mehta et al.,
2024). Ligand-decorated nanoparticles enhance receptor-mediated transcytosis
through enterocytes and M cells (Khan et al., 2023). Mucoadhesive
coatings extend epithelial contact, while thiolated and zwitterionic polymers
increase biocompatibility (Hassan et al., 2022). Incorporating
glucose-responsive elements creates a closed-loop system that mimics pancreatic
feedback (Yu
et al., 2023). Remaining challenges include reproducibility and
stability under variable intestinal conditions (Deng et al., 2025).
Advances in AI-assisted formulation modeling and recombinant design are
accelerating clinical translation (Owens, 2025).
Conclusion
The integration of recombinant technology and
nanobiotechnology offers a transformative strategy for noninvasive insulin
administration. By leveraging molecular protein engineering within advanced
nanocarrier systems, researchers are addressing the key obstacles to effective
oral delivery. Realizing the promise of swallowable insulin, however, will
require subsequent research to concentrate on scalable production methods and
robust clinical validation (Drucker, 2020).
Acknowledgment
The
authors gratefully acknowledge Dr. Naweed Imam Syed, Professor of Cell Biology,
University of Calgary, for his insight and mentorship throughout this work.
Authors’
Contribution
All
authors contributed to the conception, design, analysis, and writing of this
manuscript and approved the final version.
Data
Availability Statement
All
data supporting the findings of this study are available within the article.
References
Ahmed, S., Gupta, P. K., and Li, X. (2023).
Chitosan derivatives in biopharmaceutical nanocarriers. International
Journal of Biological Macromolecules, 241, 124521. https://doi.org/10.1016/j.ijbiomac.2023.124521
American Diabetes Association. (2024).
Standards of medical care in diabetes—2024. Diabetes Care, 47(Suppl 1),
S1–S162. https://doi.org/10.2337/dc24-SINT
Arbit, E., Kidron, M., and Arbit, E. (2022).
Noninvasive insulin systems: New directions. Diabetes Research and Clinical
Practice, 191, 109001. https://doi.org/10.1016/j.diabres.2022.109001
Chalasani, K., Russell-Jones, G. J., and Jain,
S. K. (2021). Vitamin B12-mediated oral delivery revisited. Journal of
Controlled Release, 338, 520–529. https://doi.org/10.1016/j.jconrel.2021.08.055
Choudhary, P., Ahlqvist, E., and Danne, T.
(2022). Translational aspects of oral insulin. Current Diabetes Reviews, 18(5),
489–497. https://doi.org/10.2174/1573399817666210920102515
Deng, Z., Wang, L., and Liu, Y. (2025).
AI-guided formulation modeling of oral peptides. Frontiers in Bioengineering
and Biotechnology, 13, 145212. https://doi.org/10.3389/fbioe.2025.145212
Drucker, D. J. (2020). Advances in oral peptide
therapeutics. Nature Reviews Drug Discovery, 19(4), 277–289. https://doi.org/10.1038/s41573-020-0008-4
Eldor, R., Neutel, J., and Kandeel, F. (2021).
Evaluating recombinant oral insulin analogs. Diabetes, Obesity and
Metabolism, 23(6), 1457–1463. https://doi.org/10.1111/dom.14358
Fonte, P., Araújo, F., and Sarmento, B. (2021).
Oral insulin delivery: How far are we? Journal of Diabetes Science and
Technology, 15(4), 789–804. https://doi.org/10.1177/19322968211004830
Gu, Z., Wan, T., and Li, Y. (2022).
Enzyme-nanocapsule integration for smart insulin release. ACS Nano, 16(9),
14211–14225. https://doi.org/10.1021/acsnano.2c04218
Hassan, N. A., El-Sherbiny, I. M., and Smyth,
H. D. C. (2022). Thiolated chitosan nanogels for mucosal insulin delivery. Carbohydrate
Polymers, 283, 119191. https://doi.org/10.1016/j.carbpol.2022.119191
International Diabetes Federation. (2021). IDF
diabetes atlas (10th ed.). Brussels, Belgium: Author.
Kaur, G., Narasimhulu, C. A., and Ponnusamy, T.
(2023). Lipid-based insulin encapsulation and lymphatic uptake. Nanomedicine,
46, 102681. https://doi.org/10.1016/j.nano.2023.102681
Khan, R., Wahab, A., and Mishra, A. (2023).
Intestinal receptor targeting for enhanced insulin absorption. Molecular
Pharmacology, 102(4), 356–365. https://doi.org/10.1124/molpharm.123.000678
Kulkarni, U., Mahor, A., and Singh, S. (2023).
Advances in oral insulin nanocarriers: Molecular and functional insights. Advanced
Drug Delivery Reviews, 197, 114829. https://doi.org/10.1016/j.addr.2023.114829
Lin, Y., Zhang, H., and Wang, J. (2022).
PEG-modified insulin nanoparticles for controlled uptake. Journal of
Controlled Release, 349, 281–293. https://doi.org/10.1016/j.jconrel.2022.06.061
Mehta, A., Sharma, P., and Patel, R. (2024).
Recombinant insulin folding and nano-encapsulation strategies. Biotechnology
Progress, 40(2), e3241. https://doi.org/10.1002/btpr.3241
Morishita, M., and Peppas, N. A. (2021). Is the
oral route possible for peptides? Drug Discovery Today, 26(9),
2201–2209. https://doi.org/10.1016/j.drudis.2021.06.007
Owens, D. R. (2025). Future perspectives on
insulin therapeutics. Nature Reviews Endocrinology, 21(2), 88–103. https://doi.org/10.1038/s41574-024-00990-y
Padua, D. M., Costa, E. C., and Santos, A. C.
(2023). Nano-biotechnological strategies for oral biopharmaceuticals. Biomacromolecules,
24(11), 6142–6157. https://doi.org/10.1021/acs.biomac.3c00891
Sarmento, B., Martins, J. P., and Veiga, F.
(2023). Polymer-lipid hybrids for peptide oral delivery. Biomacromolecules,
24(3), 998–1008. https://doi.org/10.1021/acs.biomac.2c01145
Yu, J., Zhang, Y., and Gu, Z. (2023).
Glucose-responsive vesicular systems for oral biotherapeutics. Proceedings
of the National Academy of Sciences of the United States of America, 120(15),
e2301121. https://doi.org/10.1073/pnas.2301121120
Zhang, Z., Liu, Y., and Wang, X. (2023).
Recombinant analog insulin delivery via a pH-sensitive hydrogel system. Journal
of Pharmaceutical Sciences, 112(5), 1345-1355. https://doi.org/10.1016/j.xphs.2023.02.015