Stomach
Cancer: Understanding Public Attention to Risk Factors, Warning Signs and Screening
Approaches, a Cross-Sectional Survey Study
Israa
Abdul Wahhab Ati
National
Cancer Institute, Cairo University
Abstract
Stomach cancer (SC) maintains constituting
a major cause of cancer-linked mortality globally and its prevalence elevates sharply,
screening of the risk factors is important. The study objected to estimate the knowledge
of risk factors especially H. pylori and warning symptoms and behavior towards
SC screening tools among the general population in an Egyptian governorate.
A cross-sectional survey was carried
on 876 participants, 512 females and 364 males in Giza city along the period from
September 2024 to December 2024 in form of self-designed questionnaire. The questions
involve personal and demographic information, the individual’s general knowledge
about SC potential risk factors, and associated signs. Another group stressed on
how likely the participants undergo screening for SC and how important the idea
of screening is? Ultimately, determine which sources of health information are the
most motivated? The demographic criteria
of 876 over 18 years of age responsive participants revealed 58.45% females and
41.55% males, 62.1% in mid-aged, 65.52% not reach college degree and 52% were working.
The most recognized risk factors were H. pylori infection, smoking and eating
a lot of salty, smoked, or pickled foods, obesity were big concerns (64.27%), (62.39%),
and (52.05%) respectively. The majority of participants (94.06%) haven’t undergone
screening despite of 85.16% of the participants believe that screening tests are
important for early diagnosis. The main mentioned reason is the cost (62.74%) followed
by fear (16.21%). The most mentioned sign linked to SC was stomach pain (75.68%),
followed by unexplained weight loss (52.05%). As expected, the main information
source was social media and internet (56%) while, healthcare providers constitute
only (3.7%). There is an extreme need to elevate the public's awareness about the
importance of early screening methods for stomach cancer and enhance trustable information
sources as health providers advising.
Keywords: Stomach cancer- risk factors- symptoms- screening-cross-sectional
study.
Corresponding author: Israa Abdul Wahhab Ati E-mail: R_rosa39@yahoo.com
Received: 29-07-2025, Accepted: 16-08-2025. Published
online: 31-08-2025
DOI: https://doi.org/10.33687/ricosbiol.03.08.72
Introduction
Statement of the Problem
Stomach cancer (SC), also termed
as gastric cancer, continues to institute a significant global health concern, and
usually ranking among the most common and lethal cancers (Bray et al., 2024).
Globally, it was recognized as the fifth most prevalent malignancy and the fifth
leading reason of cancer-associated deaths in last 5 years. Over the past decades,
increasing in the absolute number of new cases was continues to occur in spite of
decreasing in global age-standardized incidence and death rates (Li et al.,
2022). The concentrated burden observed in regions underscore the persistent impact
of region-specific risk factors such as high-sodium diets and Helicobacter pylori
(H. pylori) infection as in Egypt (Salem et al., 2019). This highlights
an enforcement need for targeted public health interferences that are susceptible
to varied regional dietary and lifestyle manners, rather than depending on a generalized
approach (Thrift and El-Serag, 2020).
In the United States, SC remains
a grave challenge, with estimated approximately 30,300 new recognition and 10,780
deaths rates in 2025. The prognosis for SC patients in the U.S. is particularly
frustrated, only 36% are related to 5-year survival but decreased to 7% when the
cancer is diagnosed at a late stage (Bray et al., 2024). Nevertheless, the prevalence
of SC in Egypt has a variant scenario as it is the 12th most noticed
cancer in both sexes representing 1.6% of the total malignancies and 2.2% cancer
death cases and 55% of cases occur between 50 and 70 years of age (Darwish et
al., 2016, Ramez et al., 2021). This wide survival variance strictly
asserts the imperative for efficient prohibition strategies and early recognition.
Extracting from epidemiological
view, a remarkable proportion of SC cases is attributed to modifiable risk factors,
which their abstractions a cornerstone of illness control (Lu et al., 2021).
The principle key modifiable factors involve alcohol consumption, H. pylori
infection, particular dietary patterns as ‘high-sodium diets and tobacco use which
is known to double the risk of stomach cancer (Boi-Dsane et al., 2023).
In spite of, scientific research
asserts that SC is largely preventable via lifestyle alterations and regular screening,
which could reduce about 60% to 70% of cases, the illness consistent to institute
a prime public health concern. So, this ongoing
issue indicates to a gap between what people know about prohibiting the disease
and actual health actions. If people believe that cancer is a disease could be preventable
but in the same time, they do not act on this belief, it proposes that the message
is not being translated into practice (Shah et al., 2020).
The Importance of the Study
Scientific Importance
This present study is scientifically substantial as, It
will provide a current, comprehensive assessment of public understanding regarding
stomach cancer's dietary risk factors, screening tools and prevention. The findings will provide empirical data to pinpoint
particular knowledge gaps, such as H. pylori detection, misunderstandings
about diet or, identifying barriers like fear of gastroscopy. This study potentiates
significant scientific importance as it will share a present and systematic assessment
to the academic literature on public comprehending of stomach cancer.
Applied Importance
The results of the current study will have a significant
impact on public health by supplying the evidence required to emerge highly targeted
educational programs. By improving public comprehending, the study can encourage
more people to join preventive behaviors, as H. pylori testing, healthier
diets, and smoking cessation. Study can also encourage earlier diagnosis by elevating
symptom awareness, an earlier detection is directly associated to better survival
rates. The data may also highlight the requirement for more education among healthcare
providers.
Objectives of the Study
Primary Objectives
Quantitative assessment of the
ongoing level of public understanding concerning dietary risk factors for stomach
cancer, high-salt foods, processed meats, low fruits and vegetables intake, and
their linked mechanisms of action in gastric carcinogenesis. Evaluation of public
awareness of rotted stomach cancer barring strategies. This encompasses understanding
the importance of Helicobacter pylori abstraction, catching specific dietary
modifications, the impact of alcohol and tobacco cessation, and the significance
of keeping a healthy body weight.
Secondary Objectives
Identifying other factors, such
as age, gender, educational achievement, and income, as well as socioeconomic factors.
Determination of the realized barriers that obstruct individuals from taking on
the preventive behaviors or looking for early detection of SC. Identifying the main
sources from which the public gets their health knowledge about stomach cancer.
Study Questions
To achieve the stated objectives,
the following specific study questions will guide the research:
What is the ongoing level of public
comprehension of dietary risk factors? and how precise is this comprehension paralleled
to scientific proof?
How aware are the people about
asserted SC prohibition approaches? including the importance of Helicobacter
pylori eradication, specific dietary guidelines, tobacco and alcohol control,
and maintaining a healthy body weight?
What are the main barriers that
impact individuals' wish to embrace stomach cancer preventive approaches or to request
early medical concern for potential signs or screening?
What are the most prevalent sources
of public about health information and how do these sources affect their understanding
of stomach cancer?
Dietary Risk Factors for Stomach
Cancer
Dietary factors play a crucial
role in the etiology of SC, with extensive epidemiological proof associating particular
consumption patterns to illness risk. Understanding these links is pivotal for efficient
public health prevention strategies (Richa et al., 2022).
High Salt Intake and Salt-Preserved
Foods
The increased risk of stomach cancer
is strongly linked with consumption of high-salt foods and those preserved with
salt because of direct damage to the gastric mucosal cells. High salt concentrations
can disrupt the guard mucosal barrier, resulting in inflammation, altered mucin
production, and cellular damage. This involved environment elevates the susceptibility
of the gastric lining to carcinogens and eases the colonization and activity of
a known gastric carcinogen, H. pylori (Balendra et al., 2023).
Research has displayed a significant
rise in SC risk with higher sodium consumption, especially from processed foods,
meta-analyses have pointed a statistically remarkable 15% and 24% elevated risk
for high salt intake and consumption of salted fish respectively (Yoo et al.,
2020).
Processed Meats
Consumption of variant processed
meats, is consistently associated with an increased risk of SC, particularly non-cardia
gastric cancer, while for red meat as a direct etiology is not evident. Nitrates
and nitrites can react within the acidic environment of the stomach to form N-Nitroso
compounds (NOCs) which are potent carcinogens (Kim et al., 2019). Additionally,
high-temperature cooking approaches, frying, baking, grilling, or barbecuing meats
can produce other carcinogenic compounds, notably polycyclic aromatic hydrocarbons
(PAHs) and heterocyclic amines (HCAs) (Mark et al., 2005).
Protective Role of
Fruits and Vegetables
Conversely, the lower risk of SC
is usually linked to a diet rich in a variety of colorful fruits and vegetables,
beans, whole grains, poultry and fish because of the abundance of bioactive components
within these foods. The research suggests a synergistic impact where a healthy diet
not only directly minimizes cancer risk via its antioxidant and anti-inflammatory
characteristics but also provides a healthier gastric environment not suitable to
H. pylori-induced carcinogenesis (Łuszczki et al., 2023).
Other Dietary Factors
Notably, there are other dietary
elements implemented in SC risk, excessive or even moderate alcohol consumption,
greater body fatness and also the consumption of charred foods (Urciuoli,
2016).
Prevention of Stomach Cancer
Helicobacter pylori Eradication
H. pylori is evidently classified as a Class 1 human carcinogen,
and its removal is greatly recommended as an effective and cost-efficient policy
for minimizing gastric cancer risk, especially in asymptomatic individuals (Sokic-Milutinovic
et al., 2015). Regarding the pathogenesis of H. pylori, the pathogen
induces chronic inflammation and damage of the acid-secreting glands in the stomach,
which leads to pre-neoplastic lesions as atrophic gastritis and intestinal metaplasia.
The pathogen also outputs vacuolating cytotoxin A and cytotoxin-associated gene
A, which have direct oncogenic impacts (Kusters et al., 2006). Although eliminating
H. pylori is effective, stomach cancer can still develop, particularly if
a person already has intestinal metaplasia (IM). This indicates that there may be
an irreversible point in cancer development (Khozani et al., 2024). Therefore,
public health campaigns should not only stress early detection and eradication of
H. pylori but also emphasize the need for continued monitoring for high-risk
individuals.
Lifestyle Modifications
Several lifestyle modifications
are substantial for SC prevention. Dietary alterations are essential, rich in fruits,
vegetables, and fiber, besides limiting salted, smoked, and poorly preserved foods
intake, as well as processed and red meats. Tobacco and alcohol control are also
crucial preventive measures with maintaining a healthy body weight and joining regular
physical activity (Ko et al., 2023).
Early Detection and Screening
Early recognition is a cornerstone
of promoting SC survival rates and believed to prohibit a high percentage of cases,
actually, there is no current widely implemented standard screening technique for
SC. Recently, non-invasive techniques, as oral microbiome analysis, are considered
promising approach for early detection (Xia et al., 2022).
The disconnection between what
people know about cancer prohibition and what they actually do is a prime issue.
Numerous people don't get screened because they don't have symptoms, which expose
a substantial misunderstanding of early detection. To overcome this, prevention
efforts must address both lifestyle alterations and the psychological hurdles to
screening (Izzeddin et al., 2025).
Methods
A cross-sectional study was carried
on 876 participants, 512 females and 364 males randomly selected those deal with
public hospitals’ out-patient and clinics related to digestive and gastrointestinal
complains in Giza city. The study was conducted from September 2024 to December
2024.
A self-designed questionnaire was
asked to the public, provided in Arabic, and then translated to English for study
writing. The survey was developed on the basis of evaluating the awareness of the
general public concerning the subject of SC.
The survey began with acquiring
personal and demographic information of each applicant concerning their age, sex,
level of education and occupation. Other questions have targeted the individual’s
general knowledge about the disease, potential risk factors, and related symptoms.
Another set focused on how likely the applicants undergo screening for SC and how
important the idea of screening is? Finally, assess which sources of health information
are the most influential?
Results
Demographics data
A total of 876 responsive participants,
512 females (58.45%) and 364 males (41.55%) answered the questionnaire, only participants
over 18 years of age were eligible. The demographic breakdown displayed that 62.1%
of participants were aged between 30 and 50 years. Concerning education, nearly two third of the participants
(65.5%) not reach college degree. Working participants have constituted 52% and
most were males while 33.2% were non worker mostly females table (1).
Table
1: Demographic features of participants
|
Feature |
Number |
|
|
|
Male
|
Female
|
|
|
364 (41.55%) |
512 (58.45%) |
|
Age |
|
|
|
18-29 |
42 |
68 |
|
30-39 |
132 |
182 |
|
40-49 |
96 |
134 |
|
50-59 |
54 |
72 |
|
≥60 |
40 |
56 |
|
Education |
|
|
|
Non-
educated |
37 |
92 |
|
Main
Education |
69 |
168 |
|
Secondary
school |
73 |
135 |
|
University |
136 |
83 |
|
Higher
level |
49 |
34 |
|
Occupation |
|
|
|
Student
|
18 |
33 |
|
Employed |
264 |
191 |
|
Unemployed |
44 |
247 |
|
Retired |
38 |
41 |
General knowledge about cancer
Regarding the general knowledge
and prevalence of cancer, 60.3% of participants have a personal or family cancer history. Interestingly,
most prevalent cancers are breast in females while lung and prostate cancer in males.
On the other side, a very little percentage of participants (8.1%) hear or have
knowledge about stomach cancer specifically, most of them have university education
or more, table (2).
Table
2: Knowledge about cancer general prevalence or stomach cancer.
|
Question |
Number |
|
||
|
|
Male |
Female |
Total |
% |
|
Do
you have a personal or family history of any cancer? |
|
|
|
|
|
Yes |
223 |
305 |
528 |
60.3 |
|
No |
35 |
72 |
107 |
12.2 |
|
I
don’t know |
106 |
135 |
241 |
27.5 |
|
Do
you hear or have knowledge about stomach cancer? |
|
|
|
|
|
Yes |
48 |
23 |
71 |
8.1 |
|
No |
316 |
489 |
805 |
91.9 |
Knowledge of Risk Factors and Prevention
The survey revealed that 32 out
of 876 participants (3.6%) have suffered from a stomach cancer condition all have
a history of H. pylori. Notably, the pathogen constitutes high prevalence
rate among participants (61.87%) so it acts as the first and most substantial risk
factor for SC. Regarding other risk factors, the participants have proposed that
smoking and eating a lot of salty, smoked, or pickled foods were big concerns (64.27%)
and (62.39%) respectively, followed by obesity (52.05%). The least ones were diet
lack of fruits and vegetables (21.57%) and then alcohol drinking (4.91%). Notably,
the low percentage of alcohol consumption answer may due to the absence of information
as 72.37% of participants said ‘I Don't know’, table 3.
Table
3: Awareness about risk factors of stomach cancer.
|
Question |
|
|
|
|
How
much do you think each of the following contributes to the risk of getting stomach
cancer? |
Not
at all |
Possible |
I
Don't know |
|
Eating
a lot of salty, smoked, or pickled foods |
53 |
546 |
277 |
|
Eating
very few fruits and vegetables |
476 |
189 |
211 |
|
Smoking
cigarettes |
75 |
563 |
238 |
|
Alcohol
drink |
170 |
43 |
634 |
|
Being
overweight or obese |
245 |
456 |
175 |
|
Are
you suffered from signs or diagnosed stomach cancer? |
|
|
|
|
yes |
32 |
|
|
|
No |
325 |
|
|
|
I
don’t know |
519 |
|
|
|
Are
you diagnosed H. pylori? |
|
|
|
|
yes |
542 |
|
|
|
No |
103 |
|
|
|
I
don’t know |
231 |
|
|
Awareness of symptoms related to
stomach cancer.
This part of the survey engaged
knowledge of signs potential to stomach cancer condition. The most known sign was
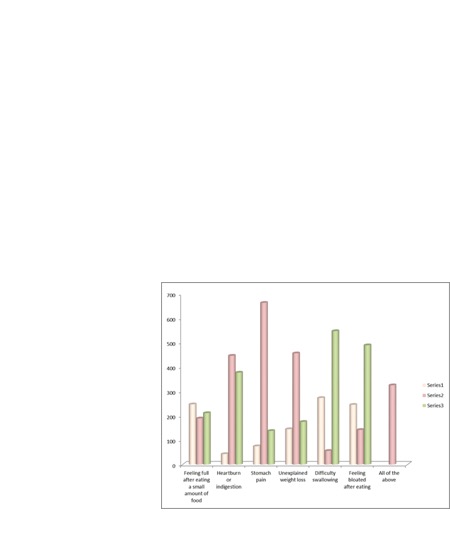
stomach pain (75.68%), followed by unexplained weight loss (52.05%), and heartburn or indigestion (50.9%). Thirty seven percent
of participants have proposed that all signs asked about are implemented and related
to stomach cancer figure 1.
Awareness about Screening and early
detection
Extracting from table (4), 85.16%
of the participants believe that screening tests can recognize stomach cancer before
signs appear, but on the contrary side, 94.06% never had a medical procedure to
screen for stomach cancer. Majority of this side think that the screening methods
are so expensive (62.74%), followed by fear of the concept (16.21%). Notably, 15.3%
of the cohort said that the screening methods were not recommended by their doctors’
figure (2).
Table
4: Knowledge about screening methods and early detection
|
Question |
Yes |
No |
|
Do
you believe that screening tests can find stomach cancer before symptoms
appear? |
746 |
47 |
|
Have
you ever had a medical procedure to screen for stomach cancer, such as a
gastroscopy? |
52 |
824 |
Figure
1: Knowledge of common symptoms related to stomach cancer.
(Series1:
Not at all Series 2: Possible Series 3: I Don't know).
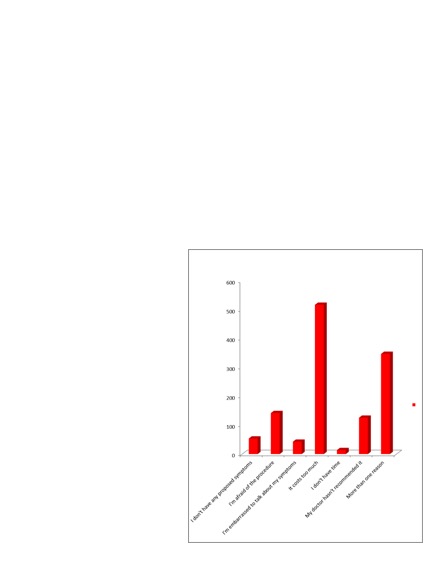
Figure 2: The reasons which prevent participants to perform
screening methods
Sources of Information
According to figure (3), 56% of
participants had mainly captured their information about cancer from social media
and internet (the most are females), persuaded by television and radio media (24.4%),
while, it was stubbed that the healthcare providers constitute only (3.7%).
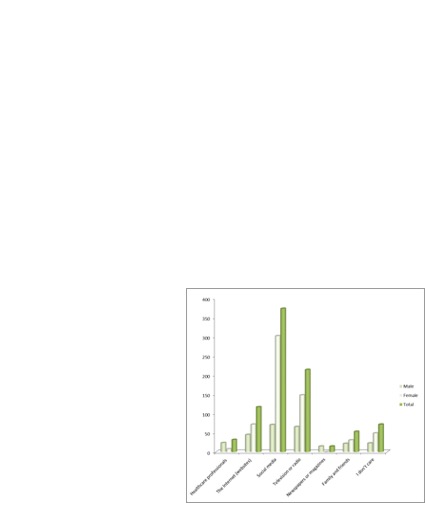
Figure
3: source of information about stomach cancer.
Discussion
Public awareness surveys display
a remarkable lack of knowledge about cancer risk factors and signs. For stomach
cancer, numerous people miscomprehend risk, this misperception misdirects prevention
efforts.
Prime barriers to early diagnosis
also subsist (Wang et al. 2024).
In current study, 60.3% of participants
have a personal or family cancer history with the most prevalent cancers are breast
in females while lung and prostate cancer in males. This finding was harmonized
with that reported about the common cancer types diagnosed in Egyptian people of
both genders (Globocan, 2020). Particularly, the little incidence of stomach cancer
mentioned in study was consistent with the Arab world cancer reports (Ibrahim and
Shash, 2022).
The study displayed that all participants
suffered from stomach cancer condition, have complained or diagnosed H. pylori.
Moreover, the finding pointed to high prevalence rate of the pathogen among participants
(61.87%).
This incidence rate is coincided
with global reports which demonstrated the common infection with H. pylori,
particularly in low- and middle-income countries as pathogen.
Also, Duan et al., (2025) stated that H. pylori influencing an estimated
more than 50% of the world's population (Grad et al. 2012).
This data asserts the major role
of H. pylori as the prime risk and causative factor in SC pathogenesis. This
is consistent with several studies,
Epidemiologic reports have displayed
that people who have chronic H. pylori infections have an elevated risk of
developing non-cardia gastric adenocarcinoma (Morais et al. 2022 and Yao
et al. 2023), as well as gastric cardia cancer (Han et al. 2023 and
Yang et al. 2021). Additionally, studies have revealed that therapeutic eradication
of H. pylori infection minimizes the risk of SC in asymptomatic individuals
(Li et al. 2019), in individuals at rosed risk due to family history (Choi
et al. 2020), and in those who have had surgery for early SC (Lee et al.
2016).
It is believed that nearly all
patients with gastric MALT lymphoma expose signs of H. pylori infection,
and the risk of emerging this cancer is fundamentally greater in affected people
than unaffected ones (Parsonnet et al. 1994 and de Sanjose et al.
2004). The strongest proof binding H. pylori infection with gastric MALT
lymphoma extracted from studies displaying that when patients with this lymphoma
are treated with antibiotics to eradicate H. pylori, their tumors shrink
(Fischbach et al. 2004 and Stathis et al. 2009).
The data extracted from current
study demonstrated other risk factors associated to SC affection, smoking then eating
a lot of salty, smoked, or pickled foods, obesity, lack of fruits and vegetables
in diet and alcohol drinking were concerns as 64.27%, 62.39%, 52.05%, 21.57% and
4.91%. These notable findings were corresponded with other previous studies, a number
of experimental investigations found that salt had a co- carcinogenic influence
when integrated with H. pylori infection. A detailed meta-analysis of longitudinal
researches exhibited that salt-rich foods have a strong induced effect on the incidence
of SC in the general population (D’Elia et al. 2014).
Also, pickled foods’ consumption
which is common dietary practice in Egypt
And also, in other regions in world
has been associated with an elevated incidence of SC in meta-analysis observational
investigations. The findings implicate that consumption of pickled vegetables/foods
may rise the risk of SC by 50% (Ren et al. 2012). Smoked food is a well-known
source of carcinogenic PAH compounds. Following to epidemiological studies, people
who regularly ate smoked-dried salted meats which contain carcinogenic heterocyclic
amines had a nearly three-fold increased risk of stomach cancer (Phukan et al.
2006).
A meta-analysis performed by Ma
et al. (2017) included ten studies revealed that consuming alcohol increases the
risk of SC. This may be attributed to the fact that alcohol can act as a solvent,
facilitating other toxic compounds to affect upper digestive tract cell lining.
This meta-analysis asserted that alcohol intake can raise the risk of SC even at
moderate levels.
Also, smoking is considered a notable
risk factor implemented in various forms of cancers, not only cancers affecting
the gastrointestinal tract (Jacob et al. 2018).
With the elevating prevalence of
obesity worldwide, findings of a study performed by Ruixin et al., (2025)
revealed that obesity significantly elevates risk of SC and remarkably affects the
treatment of patients. In the same manner, analysis of Korean wide population-based
cohort exposed SC risk was the highest in persistent obesity group, persuaded by
non-persistent obesity group compared with never-obesity set (Lim et al.,
2022).
On the other side, alteration in
dietary behavior as high fruits and vegetables intake has found to be linked to
a lower risk of cancers in over 200 epidemiological case-control and cohort studies.
The consumption of fresh vegetables and fruits, which contain antioxidant vitamins,
vitamins A and C, caused significant reduction of gastric cancer risk (Bae and Kim,
2016). A study was conducted on 14 case–control studies (5362 cases, 11,497 controls),
the findings demonstrated that high dietary vitamin C from adjusted fruit and vegetables
intake had reduced SC (Sassano et al., 2024).
The findings of present study displayed
that participants have suggested stomach pain (75.68%), unexplained weight loss
(52.05%), and heartburn or indigestion (50.9%) known as most symptoms proposed to
SC. These data were harmonized with other studies, an Omani study investigated the
recognition of symptoms of SC as “unexplained weight loss” (48.0%), “frequent bloating
and flatulence” (36.8%) and “abdominal pain” (33.8%), (Al-Azri et al. 2019). Also, Huang et al.,
(2019) mentioned that unexplained weight loss (69.5%), upper abdominal pain (69.6%) and epigastric distention (66.2%) were the most proposed symptoms
of gastric cancer.
Extracting from table (4), in spite
of, 85.16% of the participants believe that screening tests can detect stomach cancer
prior to appearance of symptoms, 94.06% of them never had a medical procedure to
screen for stomach cancer. Majority of this side think that the screening methods
are so expensive (62.74%), followed by fear of the concept (16.21%). Notably, 15.3%
of the cohort said that the screening methods were not recommended by their doctors.
Huang et al., (2019) stated
that 80.0% of study participants reported “certainly yes/probably yes” for the importance
of screening methods, parallel to the study performed by Wang et al., (2024)
who reported that a significant part of participants (78.5%) had not joined in GC
screening.
The study results which presented
in figure (3), showed that 56% of participants had primarily gained their information
about cancer from social media and internet (the most are females), followed by
television and radio media (24.4%), while, unfortunately the healthcare providers constitute only
(3.7%). Wang et al., (2024) demonstrated that videos on social media platforms
can aid the public knowledge about symptoms, etiologies, causations and treatment
methods of SC. However, both videos’ content and quality are inadequate.
Conclusion
and recommendation
General public knowledge about
stomach cancer is poor, several challenges and barriers were existed, low education,
low income, extraction the information from non-trustable sources as social media.
The link between H. pylori infection and GC is highly evident. Early understanding
of SC related symptoms and necessity of performing the screening methods for detection
of H. pylori and early diagnosis of stomach cancer to elevate the rate of
curing and survival. Encourage of doctor and other health providers to give proper
consultant for population.
References
Ahmed Ramez A., Maha El-Zaafarany, Hayam Ghazy, Refky B.,
Amany Hassan and Manar Hamed (2021). Early onset gastric cancer in Egyptian patients:
Is it really a different clinical entity? IJCBR, 5(2), 173-180.
Al-Azri M., Al-Kindi J., Al-Harthi T., Al-Dahri M., Panchatcharam
S.M., and Al-Maniri A. (2019). Awareness of Stomach and Colorectal Cancer Risk Factors.,
Symptoms and Time Taken to Seek Medical Help Among Public Attending Primary Care
Setting in Muscat Governorate., Oman. J Cancer Educ. 34(3),423-434.
Bae J.M., and Kim
E.H. (2016). Dietary intakes of citrus fruit and risk of gastric cancer incidence:
an adaptive meta-analysis of cohort studies. Epidemiol Health., 38,e2016034.
Balendra V., Amoroso C., Galassi B., Esposto J., Bareggi
C., Luu J., Scaramella L., and Ghidini M. (2023). High-Salt Diet Exacerbates H.
pylori Infection and Increases Gastric Cancer Risks. J. Pers. Med. 28,
13(9),1325.
Boi-Dsane N.A.A., Amarh V., Tsatsu S.E., Bachelle S.V.,
Bediako-Bowan A.A.A., Koney N.K., and Dzudzor B. (2023). Cross-Sectional Study for
Investigation of the Association Between Modifiable Risk Factors and Gastrointestinal
Cancers at a Tertiary Hospital in Ghana. Cancer Control. 30,10732748231155702.
Bray F., Laversanne M., Sung H., and et al. (2024). Global
cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide
for 36 cancers in 185 countries. Cancer J. Clin. 74(3),229–63.
Choi I.J., Kim C.G., Lee J.Y., and et al. (2020). Family
history of gastric cancer and Helicobacter pylori treatment. New England
Journal of Medicine 382(5),427–436.
D’Elia L., and et al. (2014). Dietary salt intake
and risk of gastric cancer. In: Zappia V., et al., editors. Advances in nutrition
and cancer, vol. 159. Berlin Heidelberg, Springer, p. 83–95.
Darwish H., Amr, S., Wedad, B., and Ahmed, Gh. (2016).
10 years’ experience in the treatment of gastric cancer: A single Egyptian Cancer
Center (NEMROCK). Pan Arab Journal of Oncology, 9 (3), 35-41.
de Sanjose S., Dickie A., Alvaro T., and et al.
(2004). Helicobacter pylori and malignant lymphoma in Spain. Cancer Epidemiology,
Biomarkers & Prevention, 13(6),944–948.
Duan Y., Xu Y., Dou Y., and Xu D. (2025). Helicobacter
pylori and gastric cancer: mechanisms and new perspectives. J. Hematol. Oncol.
23, 18(1),10.
Fischbach W., Goebeler-Kolve M.E., Dragosics B., Greiner
A., and Stolte M. (2004). Long term outcome of patients with gastric marginal zone
B cell lymphoma of mucosa associated lymphoid tissue (MALT) following exclusive
Helicobacter pylori eradication therapy: Experience from a large perspective
series. Gut, 53(1),34–37.
Gastric Cancer and Obesity: (2025). Disease Occurrence
and Perioperative Treatment. The Journal of Nutrition, 7.
Global Cancer Observatory (Globocan), Egypt fact sheet.
2020. https://gco.iarc.fr/today/data/factsheets/populations/818-egypt-fact-sheets.pdf
Grad Y.H., Lipsitch M., and Aiello A.E. (2012). Secular
trends in Helicobacter pylori seroprevalence in adults in the United States:
Evidence for sustained race/ethnic disparities. American Journal of Epidemiology,
175(1),54–59.
Han Z., Liu J.,
Zhang W., and et al. (2023). Cardia and non-cardia gastric cancer risk associated
with Helicobacter pylori in East Asia and the West: A systematic review,
meta-analysis, and estimation of population attributable fraction. Helicobacter
16,e12950.
Huang Z., Liu W., Marzo RR., Hu Z., Wong LP., and Lin Y.
(2022). High-risk population's knowledge of risk factors and warning symptoms and
their intention toward gastric cancer screening in Southeastern China. Front
Public Health, 10,974923.
Ibrahim, A.H., and Shash, E. (2022). General Oncology Care
in Egypt. In: Al-Shamsi, H.O., Abu-Gheida, I.H., Iqbal, F., Al-Awadhi, A. (eds)
Cancer in the Arab World. Springer, Singapore.
Izzeddin A Bdair, Gladys M L Maribbay, Ola A Bdair, and
et al. (2025). Awareness of
cancer risk factors, protective factors, symptoms and barriers to seek medical advice
among adult population. Asian Pac J. Cancer Prev., 26 (7), 2345-2352
Jacob L., Freyn M., Kalder M., Dinas K., and Kostev K.
(2018). Impact of tobacco smoking on the risk of developing 25 different cancers
in the UK: a retrospective study of 422,010 patients followed for up to 30 years.
Oncotarget., 9(25),17420-17429.
Kim S.R., Kim K., Lee S.A., Kwon S.O., Lee J.K., Keum N.,
and Park S.M. (2019). Effect of Red, Processed, and White Meat Consumption on the
Risk of Gastric Cancer: An Overall and Dose⁻Response Meta-Analysis. Nutrients,
11(4),826.
Ko K.P. (2024). Risk Factors of Gastric Cancer and Lifestyle
Modification for Prevention. J. Gastric Cancer, 24(1),99-107.
Kusters J.G., van Vliet A.H., and Kuipers E.J. (2006).
Pathogenesis of Helicobacter pylori infection. Clin. Microbiol. Rev., 19(3),449-90.
Lee Y.C., Chiang T.H., Chou C.K., and et al. (2016).
Association between Helicobacter pylori eradication and gastric cancer incidence:
A systematic review and meta-analysis. Gastroenterology ,150(5),1113–1124.e5.
Li F., Hu Y., Guo C., and et al. (2022). Economic
burden conferred by Population-Level Cancer screening on Resource-Limited communities:
lessons from the ESECC trial. Front Oncol., 12
Li W.Q., Zhang J.Y., Ma J.L., and et al. (2019).
Effects of Helicobacter pylori treatment and vitamin and garlic supplementation
on gastric cancer incidence and mortality: Follow-up of a randomized intervention
trial. BMJ , 366,l5016.
Lim J.H., Shin C.M., Han K.D., Lee S.W., Jin E.H., Choi
Y.J., Yoon H., Park Y.S., Kim N., and Lee D.H. (2022). Association between the Persistence
of Obesity and the Risk of Gastric Cancer: A Nationwide Population-Based Study.
Cancer Res Treat., 54(1),199-207.
Lu L., Mullins C.S., Schafmayer C., Zeißig S., and Linnebacher
M. (2021). A global assessment of recent trends in gastrointestinal cancer and lifestyle-associated
risk factors. Cancer Commun (Lond), 41(11),1137-1151.
Łuszczki E., Boakye F., Zielińska M., Dereń K., Bartosiewicz
A., Oleksy Ł., and Stolarczyk A. (2023). Vegan diet: nutritional components, implementation,
and effects on adults' health. Front Nutr., 9, 10,1294497.
Ma K., Baloch Z., He T-T., and Xia X. (2017). Alcohol consumption
and gastric cancer risk: a meta-analysis. Med Sci Monit., 23,238–46.
Mark G., Knize James S., Felton James and Felton S. (2005).
Formation and Human Risk of Carcinogenic Heterocyclic Amines Formed from Natural
Precursors in Meat. Nutrition Reviews, 63(5),158-65
Morais S., Costa A., Albuquerque G., and et al. (2022).
"True" Helicobacter pylori infection and non-cardia gastric cancer,
A pooled analysis within the Stomach Cancer Pooling (StoP) Project. Helicobacter,
27(3),e12883.
Parsonnet, J., Hansen S., Rodriguez L., and et al.
(1994). Helicobacter pylori infection and gastric lymphoma. New England
Journal of Medicine, 330(18),1267–1271.
Phukan R.K., Narain K., Zomawia E., Hazarika N.C., and
Mahanta J. (2006). Dietary habits and stomach cancer in Mizoram. India J. Gastroenterol.,
41(5),418–24.
Ren J-S., and et al. (2012). Pickled food and risk of gastric
cancer—a systematic review and meta-analysis of English and Chinese literature.
Cancer Epidemiol Biomarkers Prev., 21(6),905–15.
Richa, Sharma, N. and Sageena, G. (2022). Dietary factors
associated with gastric cancer - a review. Transl. med. commun., 7,
7.
Ruixin Xu., Siwei Pan, Yanqiang Zhang, Can Hu, Zhiyuan
Xu, Salem E., Sakr A., Younis F., and Mohamed A. (2019). "Prevalence of
Helicobacter pylori infection among farmers and non-farmers with dyspepsia".
Egyptian Journal of Occupational Medicine, 43, (2), 229-244.
Sassano M., Seyyed Salehi M.S., Collatuzzo G., Pelucchi C., and et al.
(2024). Dietary intake of vitamin C and gastric cancer: a pooled analysis within
the Stomach cancer Pooling (StoP) Project. Gastric Cancer, 27(3),461-472.
Shah SC., Nunez H., Chiu S., Hazan A., Chen S., Wang S.,
Itzkowitz S., and Jandorf L. (2020). Low baseline awareness of gastric cancer risk
factors amongst at-risk multiracial/ethnic populations in New York City: results
of a targeted, culturally sensitive pilot gastric cancer community outreach program.
Ethn. Health, 25(2),189-205.
Shirani M., Shariati S., Bazdar M., Sojoudi Ghamnak F.,
Moradi M., Shams Khozani R., Taki E., Arabsorkhi Z., Heidary M., and Eskandari DB.
(2024). The immunopathogenesis of Helicobacter pylori-induced gastric cancer:
a narrative review. Front Microbiol., 15, 1395403.
Sokic-Milutinovic A., Alempijevic T., and Milosavljevic
T. (2015). Role of Helicobacter pylori infection in gastric carcinogenesis: Current
knowledge and future directions. World J. Gastroenterol., 21(41),11654-72.
Stathis A., Chini C., Bertoni F., and et al. (2009).
Long-term outcome following Helicobacter pylori eradication in a retrospective
study of 105 patients with localized gastric marginal zone B-cell lymphoma of MALT
type. Annals. of Oncology, 20(6),1086–1093.
Thrift AP., and El-Serag HB. (2020). Burden of gastric
cancer. Clin Gastroenterol Hepatol. 18(3),534–42.
Urciuoli B. (2016). Alcohol, cured meats, and obesity play a role in stomach cancer,
study finds. Gastrointestinal Cancers, (8),1
Wang M., Yao N., Wang J., Chen W., Ouyang Y., and Xie C.
(2024). Bilibili, TikTok, and YouTube as sources of information on gastric cancer:
assessment and analysis of the content and quality. BMC Public Health, 2,
24(1),57.
Wang Q., He X.C., Geng L.X., Jiang S.L., Yang C.J., Xu
K.Y., Shen S.F., Cao W.W., Qi W., and Zhao S.P. (2024). Public awareness of gastric
cancer risk factors and screening behaviors in Shijiazhuang, China: A community-based
survey. PLoS One, 19(10),e0311491.
Xia J.Y., and Aadam A.A. (2022). Advances in screening
and detection of gastric cancer. J. Surg. Oncol., 125(7),1104-1109.
Yang L., Kartsonaki C., Yao P., and et al. (2021).
The relative and attributable risks of cardia and non-cardia gastric cancer associated
with Helicobacter pylori infection in China, A case–cohort study. The
Lancet. Public Health, 6(12),e888–e896.
Yao P., Kartsonaki
C., Butt J., and et al. (2023). Helicobacter pylori multiplex serology
and risk of non-cardia and cardia gastric cancer: A case–cohort study and meta-analysis.
International Journal of Epidemiology, dyad007.
Yoo J.Y., Cho H.J., Moon S., Choi J., Lee S., Ahn C., Yoo
K.Y., Kim I., Ko K.P., Lee J.E., and Park S.K. (2020). Pickled Vegetable and Salted
Fish Intake and the Risk of Gastric Cancer: Two Prospective Cohort Studies and a
Meta-Analysis. Cancers (Basel), 12(4),996.