Bacteriophage Therapy: A Resurgent Alternative
in the Era of Antibiotic Resistance
*Abouelhag H. A.
*Department
of Microbiology and Immunology, National Research Centre, Dokki, Giza, Egypt,
12622.
*Corresponding author: Prof. Abouelhag H. A. (Email: drabouelhag5@gmail.com)
Received: 25-05-2025,
Accepted: 20-06-2025, Published
online: 23-06-2025
DOI: https://doi.org/10.33687/ricosbiol.03.06.64
Abstract
Phage therapy, the use of bacteriophages to combat bacterial
infections, is experiencing a significant resurgence driven by the escalating crisis of antibiotic
resistance. This review provides a
comprehensive overview of the evolution of phage therapy, from its early 20th-century origins and subsequent
decline to its current status as a promising
alternative or adjunct to conventional antibiotics. We examine the
fundamental mechanisms of phage action,
highlighting their specificity for bacterial targets and their lytic
capabilities against even multidrug-resistant strains, while often sparing the
host microbiota. Current applications
are explored across various domains, including the treatment of chronic and resistant infections in humans,
personalized medicine approaches,
veterinary uses, and food safety applications. Key innovations, fueled by advances in genomics and synthetic
biology, such as phage engineering, cocktail
formulations, phage-derived enzymes
(e.g., endolysins), and novel delivery
systems, are discussed as crucial enhancers of
therapeutic potential. Despite its promise, phage therapy faces significant challenges, including complex regulatory
pathways, manufacturing and
standardization hurdles, the potential for bacterial resistance to phages, and host immune responses.
Addressing these limitations through rigorous
clinical trials, standardized protocols, and continued research is
essential. This review underscores the critical need to integrate
phage therapy into modern medical
paradigms as a vital tool in the global fight against
antibiotic-resistant infections, outlining future directions for research and clinical implementation.
Keywords: antibiotic resistance, bacteriophages, phage therapy, clinical
applications, genetic engineering, innovations, multidrug-resistant bacteria.
Introduction
The escalating crisis
of antibiotic resistance (AMR) represents one of the most significant global health threats of the 21st century. Decades of
widespread, and often inappropriate, use of antibiotics in human medicine,
veterinary practice, and agriculture have driven the selection and proliferation of bacteria resistant to multiple drugs, rendering previously effective
treatments obsolete (World Health Organization, n.d.). Common infections are becoming
increasingly difficult, and sometimes
impossible, to treat, leading to prolonged illness, increased
mortality rates, and substantial economic burdens on healthcare systems worldwide.
The World Health Organization (WHO) has repeatedly warned that without urgent, coordinated action, the
world is heading towards a post-antibiotic era where common infections and minor injuries could once again prove fatal. This alarming trajectory underscores the
critical need for innovative strategies and alternative therapeutic agents to combat bacterial infections, particularly those caused
by multidrug-resistant (MDR) pathogens.
Amidst this challenge, there is a renewed and
rapidly growing interest in a therapeutic approach that predates the antibiotic era: bacteriophage therapy. Bacteriophages, often simply
called phages, are viruses that naturally infect and kill bacteria. Discovered independently by Frederick Twort in 1915
and Félix d’Hérelle in 1917 (Summers, 1999), these bacterial predators are the most abundant biological
entities on Earth, playing crucial
roles in shaping microbial ecosystems. Phages possess remarkable specificity, typically targeting only particular
strains or species of bacteria. This high degree of specificity is a key advantage of phage therapy; unlike broad-spectrum antibiotics
which can disrupt the host's
beneficial microbiota (leading to dysbiosis and secondary infections like Clostridioides difficile), phages can selectively eliminate pathogenic bacteria while leaving the commensal flora
largely undisturbed (Górski et al., 2016; Sulakvelidze et al., 2001). Furthermore, phages can
replicate exponentially at the site of infection
as long as susceptible host bacteria are present, essentially acting as self- amplifying drugs, and they possess diverse
mechanisms to overcome
bacterial defenses.
The concept of using phages therapeutically was
pioneered by d'Hérelle shortly after their
discovery. He demonstrated their potential by successfully treating bacterial dysentery
and later applied them against other infections
like cholera and typhoid fever during
the 1920s (Summers, 1999). Phage therapy gained considerable traction, particularly in Eastern Europe and the
former Soviet Union, where institutions like the Eliava Institute in Tbilisi, Georgia, became centers for phage
research and application, continuing this practice even through the antibiotic age (Górski et al., 2016). However, in the Western world, the advent of penicillin
and subsequent broad-spectrum antibiotics in the
1940s overshadowed phage therapy (Principi et al., 2019). The perceived reliability, ease of use, and broad applicability of antibiotics, coupled with methodological shortcomings in some early
phage therapy studies (lack of rigorous controls,
poor characterization of phage preparations, limited accessibility of research published
in non-English journals), led to its decline in most parts of the world (Principi et al., 2019; Summers, 1999).
Today, facing the stark reality of dwindling
antibiotic efficacy, the scientific and medical communities are revisiting phage therapy with renewed vigor.
Driven by the urgent need for alternatives to combat AMR,
modern research is leveraging advances in genomics, molecular biology, and synthetic biology to overcome the historical limitations of phage therapy
and unlock its full potential (Hatfull et al., 2022).
Contemporary studies are exploring the use of naturally occurring phages, precisely
characterized phage cocktails, and genetically engineered phages to treat a wide range
of challenging infections caused by MDR bacteria.
This review aims to provide a comprehensive
overview of the evolution, current status, and
future prospects of bacteriophage therapy as a viable alternative and adjunct
to conventional antibiotic
treatments. We will delve into the fundamental mechanisms of phage action, explore their diverse applications in clinical settings,
veterinary medicine, and food safety,
and discuss the cutting-edge innovations shaping the field.
Furthermore, we will critically examine the
challenges and limitations that must be addressed
– including regulatory hurdles, manufacturing complexities, potential for phage resistance, and host immune
responses – to facilitate the successful integration of phage therapy
into mainstream medical practice. By synthesizing the historical context, recent advancements, and ongoing research,
this review seeks to highlight the significant potential of phage therapy
to contribute to the global
fight against antibiotic resistance and transform the management of bacterial
infections.
Mechanisms of Action
The therapeutic efficacy of bacteriophage
therapy hinges on the intricate biological mechanisms governing
phage-bacteria interactions and the subsequent
response within the host environment. Understanding these mechanisms
is fundamental to optimizing phage selection, administration
strategies, and predicting treatment outcomes
(Sulakvelidze et al., 2001). Phages employ sophisticated
strategies to infect, replicate within, and ultimately destroy
their specific bacterial targets.
Central to phage activity are their distinct
life cycles, primarily the lytic and lysogenic
cycles. The lytic cycle represents the aggressive, bacteria-killing phase most relevant
for direct therapeutic
action. In this cycle, a lytic phage first adsorbs to a susceptible bacterium by recognizing and binding to
specific receptors on the bacterial cell surface (e.g., lipopolysaccharides, outer membrane proteins, pili,
flagella). This binding event triggers
the injection of the phage's genetic material (DNA or RNA) into the bacterial cytoplasm. Once inside, the phage genome
hijacks the host cell's machinery, redirecting
it towards the replication of phage DNA/RNA and the synthesis of phage
structural components (capsid
proteins, tail fibers, etc.). Crucially, lytic phages often produce enzymes like endolysins and holins late in
the cycle. Holins create pores in the bacterial cytoplasmic membrane, allowing
endolysins access to the peptidoglycan layer of the cell
wall, which they degrade. This enzymatic breakdown weakens the cell wall,
leading to osmotic lysis – the
rupture of the bacterial cell – releasing hundreds of newly assembled progeny phages (Cahill and
Young, 2019; Young, 1992). These
newly released virions can then infect surrounding susceptible bacteria, amplifying
the antibacterial effect locally. This rapid replication and bacterial killing
make lytic phages the preferred
candidates for treating
acute infections (see Figure 1).
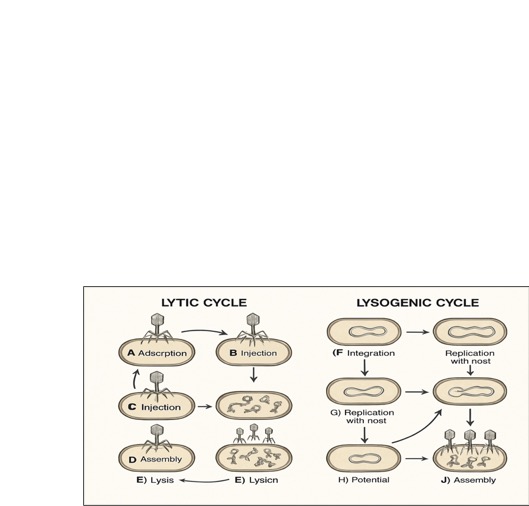
Figure 1: Comparison of lytic and lysogenic bacteriophage life cycles.
In contrast, the lysogenic cycle involves the integration of the phage genome (now termed a prophage) into the host
bacterium's chromosome, or its maintenance as an extrachromosomal plasmid. The prophage replicates passively
along with the bacterial DNA during
cell division, without immediately harming the host. Bacteria carrying a prophage are termed lysogens. This state
can persist for many generations, potentially
conferring new properties to the bacterium, such as resistance to
infection by similar phages (superinfection immunity) or even the production of virulence factors (lysogenic conversion, e.g., diphtheria toxin,
cholera toxin). Under certain environmental triggers (e.g., UV radiation, chemical stress, nutrient depletion), the
prophage can excise itself from the
bacterial chromosome and enter the lytic cycle, leading to phage replication and lysis (Howard-Varona et al., 2017). While lysogenic phages
are generally avoided
for direct therapy due to the
potential for lysogenic conversion and the lack of immediate
bacterial killing, their study provides insights into phage-bacteria
co-evolution, and engineered temperate phages might hold future therapeutic potential.
The remarkable host specificity of phages is a defining characteristic and a
major therapeutic advantage.
This specificity is primarily determined by the initial adsorption
step, requiring a precise molecular match between phage attachment structures
(e.g., tail fibers) and specific
bacterial surface receptors (Labrie et al.,
2010). This lock-and-key mechanism ensures that a given phage typically infects only a
narrow range of bacterial strains or
species, leaving non-target bacteria, including beneficial members of the host microbiota, unharmed. This contrasts
sharply with the collateral damage often caused by broad-spectrum antibiotics (see Figure 2).

Figure 2: Bacteriophage host specificity. Only phages with matching receptors
can bind and infect the bacterial cell.
However, the interaction is not solely dictated
by the phage. Bacteria have evolved numerous resistance mechanisms to
evade phage predation. These include modifying
or masking surface receptors to prevent phage adsorption, producing
extracellular matrices that block
phage access, deploying restriction-modification systems that degrade foreign DNA upon injection, and utilizing CRISPR-Cas adaptive
immune systems to recognize and cleave phage
genetic material based
on previous encounters (Labrie et al., 2010). The dynamic co-evolutionary
arms race between phages and bacteria means
that bacterial resistance to specific phages can emerge, necessitating
strategies like using phage cocktails
(mixtures of different phages targeting the same bacterium via different
receptors or mechanisms) to mitigate this risk (Chan
et al., 2013)
(see Figure 6).
Finally, the interaction between phages and the
host immune system adds another layer
of complexity. When introduced therapeutically, phages can be recognized as foreign entities, potentially triggering
innate and adaptive immune responses. The production
of neutralizing antibodies against phages can lead to their rapid clearance from circulation, potentially limiting the efficacy
of systemic phage therapy, especially
upon repeated administration (Hodyra-Stefaniak et al.,
2015). The extent
of this immune
response depends on factors like the phage type, dosage, route of
administration, and the host's immune
status. While often viewed as a hurdle, the immune response is not always detrimental; in some cases,
phage-induced bacterial lysis can release bacterial antigens and pathogen-associated molecular patterns (PAMPs) that
stimulate a beneficial host immune response
against the infection. Furthermore, some phages
have evolved mechanisms to
persist despite host immune responses, such as encapsulation or adaptation within the host environment, enhancing
their ability to evade immune detection
and prolong their activity (Hodyra-Stefaniak et al., 2015). Understanding and potentially modulating these phage-immune
interactions is crucial for developing effective and safe phage therapy protocols.
Current Clinical Applications
Phage therapy is increasingly being
recognized and explored
as a viable clinical strategy, particularly for tackling
challenging bacterial infections that are refractory to
conventional antibiotic treatments. Its
applications span various medical fields, demonstrating significant potential, although widespread adoption is still hindered by regulatory and logistical challenges. The primary driver for its clinical
resurgence is the urgent need to address
infections caused by multidrug-resistant (MDR) bacteria, where
treatment options are severely limited. One of the most notable uses of modern phage therapy is in managing chronic
and persistent infections, especially those involving biofilms,
which are notoriously difficult for
antibiotics to penetrate and eradicate. Phage therapy has shown promise in treating chronic wounds, osteomyelitis (bone infections), prosthetic joint infections, and chronic respiratory infections in patients with
conditions like cystic fibrosis (Fabijan et al., 2020) (see Figure
3).
For instance, successful case reports For instance, successful case reports detail the use of phage therapy, often in combination with
antibiotics, to resolve long-standing infections caused by MDR pathogens like Pseudomonas aeruginosa and
Staphylococcus aureus. A case involving a Siamese
cat with a surgical wound infected by multidrug- resistant P. aeruginosa demonstrated
complete healing after 14 weeks following treatment
with a combination of a specific phage and antibiotics, underscoring the potential
in both human and veterinary medicine (Fabijan et al., 2020).
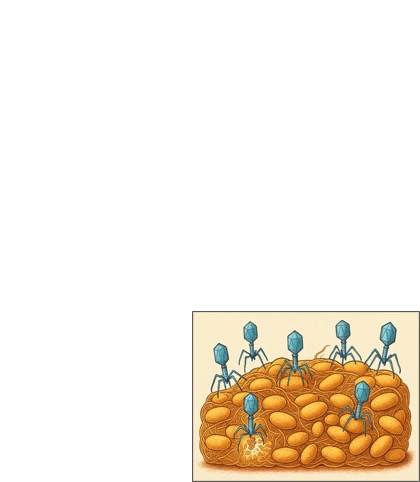
Figure 3: Bacteriophages penetrating a bacterial biofilm
and lysing embedded
bacteria.
Phage therapy is particularly suited
for personalized medicine
approaches. Given the high
specificity of phages, treatment often involves
identifying the specific bacterial strain causing the infection and
then selecting or isolating phages that are effective against that particular strain. This tailored approach enhances
therapeutic effectiveness and minimizes disruption to the patient's beneficial microbiota (Pirnay et al., 2011).
Several centers, particularly in countries with a longer
history of phage use like Georgia and Poland, as well as emerging programs
in the US, Belgium, and Australia, offer compassionate
use or experimental phage therapy. These programs often involve creating customized phage preparations
(sometimes cocktails of multiple phages) for
patients with life-threatening or debilitating infections
unresponsive to standard
care. While often conducted
outside large-scale randomized controlled trials (RCTs), these compassionate use cases provide valuable
real-world evidence and case reports documenting both successes and challenges.
Clinical trials investigating phage therapy are gradually increasing in number and
rigor, although they still lag
behind those for conventional drugs. Early trials and ongoing studies are evaluating the safety and efficacy
of phage preparations for various conditions, including
urinary tract infections, diabetic foot ulcers,
burn wound infections, and respiratory infections. For example,
standardized phage cocktails targeting E.
coli, P. aeruginosa, and S. aureus have been tested. While some
trials have shown promising results
regarding safety and bacterial load reduction, demonstrating definitive clinical superiority over
standard care in large RCTs remains a key objective and challenge (Jault et al., 2019). Regulatory pathways, such as the FDA's compassionate use programs, allow access for some patients, but broader
approval requires more extensive clinical
validation.
Different routes of administration are employed
depending on the site and type of infection. Topical application is common for wound infections
and skin conditions. Oral administration is used for gastrointestinal infections
or potentially for systemic effects,
although phage stability in the gut environment can be a concern. Intravenous administration allows for systemic
delivery to treat bloodstream infections or deep- seated infections like osteomyelitis. Aerosolized phage delivery
systems are being explored for treating respiratory infections, allowing for
localized treatment while minimizing systemic
exposure (Malik et
al., 2017). The optimal route
and dosing regimen
often need to be determined on a case-by-case basis.
The specific
bacterial targets most commonly addressed in recent clinical applications reflect
the major AMR threats, including
ESKAPE pathogens (Enterococcus faecium,
Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species). Tailored phage
formulations and cocktails are being developed and tested
against these critical pathogens (Jault et al., 2019; Rhoads et
al., 2009). While the
clinical application of phage therapy is still evolving, the accumulating evidence
from case studies,
compassionate use programs, and initial clinical
trials provides a strong rationale
for its continued development as a crucial
tool against antibiotic-resistant infections.
Applications
Beyond Human Medicine
The potential of bacteriophage therapy extends
significantly beyond human clinical applications,
offering promising solutions in veterinary medicine, food safety, and agriculture, primarily driven by the need to reduce antibiotic use and combat resistance in these sectors
as well.
In veterinary medicine, phage therapy is increasingly
considered a viable alternative or adjunct to antibiotics for treating
infections in both livestock and companion
animals (Jończyk-Matysiak et al., 2021). Antibiotic resistance is a growing concern in animal health, impacting treatment efficacy and potentially contributing to the pool of resistant bacteria that can affect humans. Phage therapy has shown effectiveness in
managing conditions such as mastitis
in cattle, salmonellosis in poultry, and respiratory diseases in pigs. Studies indicate that phage
treatments can significantly reduce bacterial loads and prevent disease, enhancing animal health while decreasing
reliance on antibiotics (Jończyk-Matysiak
et al., 2021). Personalized approaches, similar to those in human medicine, are also applicable. For
instance, specific phages have been applied topically to successfully treat antibiotic-resistant skin infections in
dogs, demonstrating how customization
can improve outcomes and reduce
collateral damage to beneficial bacteria (Pirnay et al., 2011). The use of phages
in veterinary settings aligns with the 'One
Health' approach, recognizing the interconnectedness of human, animal, and environmental health
in tackling AMR.
Food safety represents another major area where phages hold
considerable promise. Phages can be
used to specifically target and eliminate pathogenic bacteria that contaminate food products, thereby improving safety and potentially extending shelf life (Endersen
et al., 2014). Phage preparations have been approved
by regulatory agencies
like the FDA and USDA (and in the EU) for use as food processing aids,
particularly against pathogens like Listeria monocytogenes on ready-to-eat
meat and poultry products
(e.g., Bacteriophage P100) (Goodridge and Abedon, 2003). Research has demonstrated
that phage treatments can effectively reduce the presence of pathogens such as Salmonella and
E. coli in various
food items, including fresh produce, meats,
and dairy products.
By incorporating phage treatments into food production and processing protocols
(e.g., spraying onto carcasses or adding to packaging), producers can mitigate the risks associated with bacterial
contamination, offering a natural and targeted
biocontrol method (Endersen et al., 2014) (see Figure 4).

Figure 4: Application of bacteriophages in food safety to reduce
pathogens on food products.
Challenges in this area Challenges in this area
include ensuring phage survival and activity in complex food matrices and varying environmental conditions (Goodridge and Abedon, 2003).
In agriculture, phages are being explored as
biocontrol agents to combat bacterial diseases
in plants. Phytopathogenic bacteria cause significant crop losses worldwide, and resistance to traditional bactericides
is emerging. Phages that specifically target
plant pathogens, such as Xanthomonas
species (causing blights and spots) or Pseudomonas syringae, offer an
environmentally friendly alternative to chemical treatments (Jones et al., 2007). Field trials are
investigating the efficacy of phage applications
in controlling diseases in various crops. Key challenges include ensuring phage stability and persistence in the
agricultural environment (phyllosphere, rhizosphere)
under fluctuating conditions like UV radiation and desiccation (Jones et
al., 2007).
These applications highlight
the versatility of bacteriophages as targeted antibacterial agents across diverse sectors, contributing to a broader
strategy for reducing antibiotic dependency and managing bacterial threats in interconnected ecosystems.
Innovations and Advances in Phage Therapy
Innovations and advancements in phage therapy
are significantly enhancing its efficacy and expanding its potential applications, largely driven by progress in genomics,
molecular biology, and synthetic biology. Researchers are moving beyond simply isolating
naturally occurring phages to actively engineering and optimizing them for improved therapeutic performance.
One of the most promising areas is phage
engineering. Scientists are manipulating phage genomes
to enhance desirable traits, such as improving their
stability, broadening their host range (to target more
bacterial strains), increasing their potency, or boosting their resistance to bacterial defense
mechanisms (Pires et al., 2016). Techniques like CRISPR-Cas9
gene editing are being utilized to modify phages, for example, to create phages that can effectively combat antibiotic-resistant bacteria
by directly targeting
and disrupting bacterial DNA or essential genes (Yosef et al., 2015)
(see Figure 5).
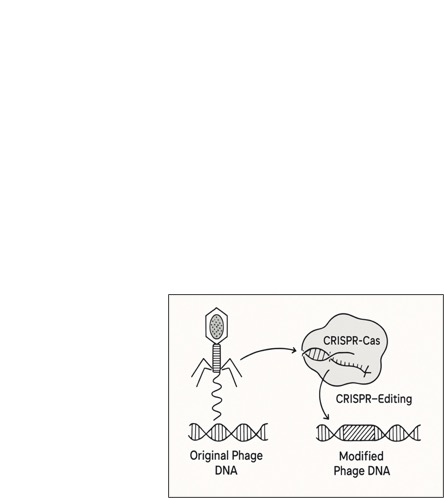
Figure 5: Conceptual
diagram of phage engineering using CRISPR-Cas technology
to modify phage DNA.
These engineered phages These engineered phages can be designed to overcome specific bacterial
resistance mechanisms or to express
antibacterial proteins themselves.
Phage display
technology has revolutionized the development of extensive libraries of genetically
engineered phages that can be screened for their ability to bind to specific bacterial targets (Smith, 1985).
This capability allows researchers to develop broad- spectrum phage therapies by identifying and selecting phages
targeting multiple bacterial strains
or species. Libraries with vast diversity (e.g., up to 10^10 different variants)
enable the rapid
identification of effective phages for therapeutic use.
Techniques like
biopanning, involving repeated cycles of selection and amplification, are crucial for enriching
phage clones with high binding affinity to targeted pathogens, thus enhancing the therapeutic arsenal
against bacterial infections (Pande et al.,
2010).
Beyond modifying whole phages, researchers are harnessing phage-derived products, particularly lytic enzymes like endolysins and depolymerases. Endolysins are enzymes produced
by phages late in the lytic cycle to degrade the bacterial
peptidoglycan cell wall from within, causing lysis. When
applied externally (as purified recombinant proteins),
endolysins can rapidly kill Gram-positive bacteria and, with modifications, Gram-negative bacteria as well. They offer advantages like high
specificity, low likelihood of resistance development (as they target
essential conserved structures), and the ability to kill antibiotic-resistant strains (Fischetti, 2005).
Depolymerases are enzymes found on some phages that degrade
the capsular polysaccharides or exopolysaccharides
forming bacterial biofilms, helping phages penetrate these protective layers or disrupting the biofilm structure directly.
Phage cocktails,
mixtures containing multiple distinct phages targeting the same bacterial
species (often via different receptors or lytic mechanisms), are a key strategy to combat
the emergence of phage-resistant bacterial mutants and broaden the effective host range of a therapeutic preparation (Chan
et al., 2013). By presenting bacteria with multiple simultaneous threats, cocktails make it significantly
harder for resistance to develop against
all components concurrently.
Significant innovations are also occurring in phage
delivery systems to overcome challenges
related to stability, bioavailability, and targeted delivery. Phages can be sensitive to environmental conditions
(e.g., pH in the stomach) and host immune clearance.
Encapsulation techniques using polymers, liposomes, or hydrogels can protect phages from degradation, control their release kinetics, and facilitate delivery
to specific infection
sites (Malik et al., 2017; Puapermpoonsiri et al., 2009)
(see Figure 7).
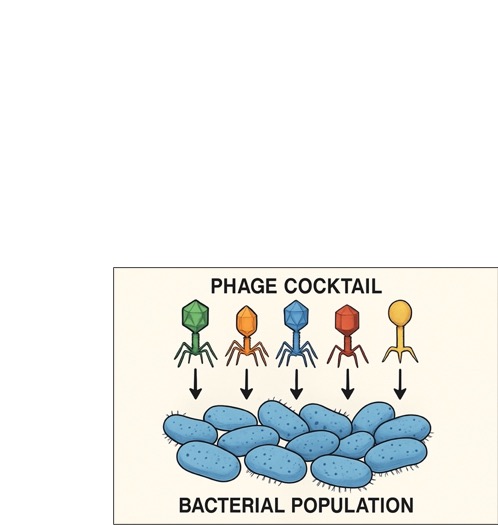
Figure 6: A phage cocktail, consisting of diverse
phages, used to target a bacterial population.
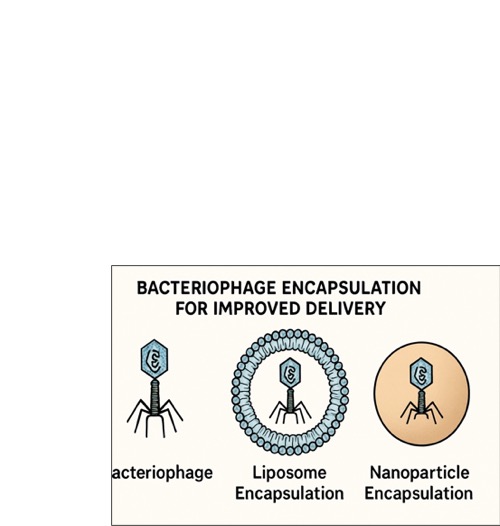
Figure 7: Encapsulation methods like liposomes
and nanoparticles protect
phages for improved delivery.
Nanoparticles
are also being explored Nanoparticles are also being explored as carriers for targeted phage delivery and even for combined diagnostic/therapeutic (theranostic) purposes (Peng and Chen, 2021). Advancements in aerosolized phage delivery systems
are being investigated for treating respiratory infections, allowing for localized treatment
while minimizing systemic
exposure (Malik et al., 2017).
Synergistic
approaches, particularly combining phages with conventional antibiotics (phage-antibiotic
synergy, PAS), are gaining considerable attention. Studies have shown that sub-lethal concentrations of
certain antibiotics can enhance phage propagation or that phages can re-sensitize antibiotic-resistant bacteria to the drug. This combination can lead to more effective
bacterial clearance, reduce the required
doses of both agents, and potentially slow the development
of resistance to both phages and antibiotics
(Comeau et al., 2007; Tagliaferri et al., 2021). This
synergistic effect is being explored in various clinical
settings, potentially leading to improved
patient outcomes, especially for difficult-to-treat infections (Chaudhry et al.,
2017).
Other emerging innovations include the
development of phage-based vaccines, using phages
as platforms for antigen display or delivery (Clark and March, 2004), and the exploration of oncolytic phages,
engineered to specifically target cancer cells
or tumor- associated bacteria (Yacoby et al., 2007). While still in early stages,
these areas highlight
the expanding versatility of phage-based technologies.
These innovations collectively aim to overcome
the limitations of natural phages and traditional antibiotics, paving the way for more effective, targeted, and sustainable antibacterial strategies.
7. Challenges and Limitations
Despite the considerable promise and renewed
interest, the widespread clinical implementation
of phage therapy faces a complex array of challenges spanning regulatory, developmental, biological, and
logistical domains. Addressing these obstacles
is crucial for successfully integrating phage therapy into mainstream medical practice (Verbeken et al., 2014).
One of the most significant hurdles is regulatory.
Unlike chemically synthesized small- molecule antibiotics, phages are biological entities capable of replication and evolution. This unique nature does not fit neatly
into existing regulatory frameworks designed for conventional pharmaceuticals. Regulatory agencies like the FDA
and EMA require rigorous approval
processes, typically involving
standardized manufacturing, preclinical safety data, and large-scale randomized controlled trials
(RCTs) to demonstrate safety and efficacy
(Verbeken et al.,
2014). Proving efficacy
for phages can be challenging given their high
specificity (requiring precise matching to the infecting bacteria) and the potential need for personalized or
adaptable phage cocktails. Early clinical studies often lacked adequate controls and employed crude preparations,
complicating the interpretation of historical data and necessitating
modern, high-quality trials (Merabishvili et
al., 2009). Establishing standardized protocols for phage
isolation, characterization,
manufacturing (ensuring purity, potency, and freedom from contaminants like bacterial toxins or
antibiotic resistance genes), and storage remains a critical need (Pirnay et al., 2011). The absence
of universally accepted quality controls and
manufacturing standards (like Good Manufacturing Practice - GMP for phages) creates
significant barriers to large-scale production and clinical use.
Manufacturing and
quality control present specific technical challenges. Producing well-characterized, high-titer phage preparations free from bacterial
debris, endotoxins, and potentially harmful phage-encoded genes (e.g., toxins, antibiotic resistance genes) requires sophisticated purification and
quality assessment methods. Ensuring the stability and maintaining the viability of phage preparations during storage and transport is also essential but can be difficult, as phages can be sensitive to physical and chemical conditions
(Pirnay et al.,
2011). Scaling up production to meet potential
clinical demand while
maintaining strict quality standards is another major logistical and economic challenge.
Biological challenges primarily revolve around bacterial
resistance to phages and host immunogenicity. Just as bacteria
evolve resistance to antibiotics, they can also develop resistance
to phages through various mechanisms (e.g., receptor modification,
CRISPR- Cas systems) (Labrie et
al., 2010). While
the use of phage cocktails can mitigate this,
the potential for resistance necessitates ongoing surveillance and the continuous discovery or engineering of new phages.
The immunogenicity of phages is another concern.
The host immune
system can recognize
phages as foreign
and mount an immune response,
primarily through antibody production,
which can lead to rapid phage
clearance and reduced therapeutic efficacy, particularly upon repeated
administration (Hodyra- Stefaniak et al., 2015; Łusiak-Szelachowska et al., 2014). While this response is not always
detrimental and can sometimes be leveraged, minimizing adverse immune reactions through careful phage selection,
purification, dosing strategies, and potentially
phage engineering is important for safety and effectiveness (Łusiak- Szelachowska et al., 2014).
Furthermore,
the narrow host range of most phages,
while advantageous for specificity, can also be a limitation. It requires
accurate and rapid diagnosis of the causative
bacterial agent and susceptibility testing to select effective phages.
This contrasts with the empirical use
often possible with broad-spectrum antibiotics. Developing rapid diagnostic tools and extensive,
well-characterized phage libraries is essential to overcome this practical challenge.
Finally, issues related
to pharmacokinetics and pharmacodynamics (PK/PD) – how phages distribute within the body, reach the infection site at
sufficient concentrations, and interact with bacteria over time – are still not fully understood and require further investigation to optimize dosing regimens and
administration routes (Malik et al., 2017). Ethical
considerations surrounding the use of self-replicating biological agents also
need careful consideration and public
discourse.
Overcoming these multifaceted challenges will
require concerted efforts from researchers,
clinicians, regulatory bodies, and industry stakeholders to develop standardized protocols, conduct rigorous
clinical trials, and establish clear pathways for the safe and effective use of phage
therapy.
8. Future Directions and Perspectives
The trajectory of phage therapy research and
development points towards an increasingly important role in combating bacterial
infections, particularly in the face of escalating antibiotic resistance. However,
realizing this potential requires addressing the current challenges and capitalizing on recent scientific and technological advancements. Several key future directions are emerging.
First and foremost
is the critical need for large-scale, rigorously designed randomized controlled
trials (RCTs). While case studies and compassionate use provide valuable anecdotal evidence, robust RCTs are
essential to definitively establish the safety and efficacy of phage therapy for specific indications compared to
standard-of-care treatments. These
trials need to address complexities such as appropriate control groups,
standardized phage preparations, defined clinical endpoints, and strategies for handling phage specificity and
potential resistance development. Generating high- quality clinical evidence
is paramount for gaining regulatory approval and acceptance by the broader medical
community.
Developing standardized protocols and regulatory pathways is another crucial
area. Collaboration between
researchers, industry, and regulatory agencies (like FDA, EMA) is needed
to establish clear guidelines for phage manufacturing (GMP standards), quality control,
characterization, preclinical testing,
and clinical trial design specifically tailored to the
unique nature of phages. Harmonizing regulations internationally would also facilitate broader development and access.
Continued advancements in phage engineering and synthetic biology
hold immense promise. Future research will likely
focus on creating phages with enhanced properties: broader host ranges, reduced immunogenicity, improved stability
and delivery characteristics, enhanced
biofilm penetration capabilities, and mechanisms to actively combat bacterial resistance. Engineering
phages to deliver specific payloads (e.g., enzymes,
toxins targeting bacteria) or to work synergistically with the host immune system are also exciting avenues.
Synthetic biology approaches may enable the de novo
design and construction of phages with precisely defined
characteristics.
Further exploration of phage-microbiome
interactions is warranted. Understanding how therapeutic phages interact with the complex microbial communities in the human body
(e.g., gut, respiratory tract) is important for predicting efficacy and
potential off- target effects.
Leveraging phages to selectively modulate the microbiome for therapeutic benefit
is an emerging field.
Optimizing phage discovery
and selection processes
is also key. Developing high- throughput methods for isolating and characterizing
phages against clinically relevant pathogens,
including MDR strains, is essential. Building extensive, well-curated phage libraries, potentially linked to rapid diagnostic tools that identify
the causative agent and its phage susceptibility profile, will
be vital for implementing personalized or readily available phage
therapy.
Improving delivery systems
to ensure phages
reach the site of infection at adequate concentrations and remain active remains
a priority. Research into advanced formulations, encapsulation methods, and targeted delivery strategies
will continue.
Finally, educating clinicians, policymakers, and the public about
the potential and limitations
of phage therapy is necessary to foster acceptance and facilitate its integration into clinical practice. Phage
therapy is unlikely to completely replace antibiotics but rather will serve as a valuable
alternative or adjunct,
particularly for difficult-to-treat infections. Its successful integration will likely
involve its use in
combination therapies (e.g., phage-antibiotic synergy) and within specific clinical
niches where antibiotics fail.
The path forward requires sustained investment
in research, interdisciplinary collaboration, and a flexible
yet rigorous approach
to regulation to translate the promise of phage therapy
into tangible clinical
benefits in the fight against
AMR.
Conclusions
The era of antibiotics, while revolutionary, is facing
an unprecedented challenge due to the global rise of antimicrobial
resistance. As conventional therapies lose their effectiveness against increasingly resilient pathogens,
bacteriophage therapy is re- emerging
from its historical roots as a highly promising and scientifically validated alternative and adjunct strategy. Its
inherent specificity allows for the targeted
elimination of pathogenic bacteria while preserving the host's beneficial microbiota, a distinct
advantage over broad-spectrum antibiotics. Furthermore, the ability of phages to self-replicate at the site of infection
and their potential to overcome existing resistance mechanisms offer unique therapeutic benefits.
Significant progress has been made in
understanding phage biology, developing methods for phage characterization and production, and exploring diverse
applications ranging from treating MDR infections in humans and animals to ensuring food safety.
Innovations in phage engineering, cocktail formulation, delivery systems, and synergistic combinations with antibiotics are continually enhancing the potential and applicability of this therapeutic modality. Modern research, leveraging genomics and
synthetic biology, is actively
addressing the historical limitations and paving the way for more potent,
reliable, and safer phage-based treatments.
Despite this progress, substantial challenges
remain. Regulatory frameworks require adaptation,
manufacturing processes need standardization and scaling, and issues like bacterial resistance to phages and host
immunogenicity must be effectively managed through ongoing
research and strategic development. Rigorous, large-scale clinical trials are essential to provide definitive
evidence of efficacy and safety, facilitating regulatory approval and broader
clinical acceptance.
In conclusion, bacteriophage therapy represents
a critical component in the multifaceted
approach required to combat the AMR crisis. While not a panacea, its unique mechanisms and adaptability offer a powerful
tool, particularly for infections where conventional antibiotics have
failed. Continued investment in research, interdisciplinary collaboration,
robust clinical validation, and the development of supportive regulatory pathways are imperative to fully realize
the potential of phages and successfully integrate them into
21st-century medicine as a vital weapon against bacterial infections.
References
Cahill, J., and Young, R. (2019).
Phage lysis: Multiple
genes for multiple
barriers. Advances in Virus Research, 103, 33–70. https://doi.org/10.1016/bs.aivir.2018.09.003
Chan, B. K.,
Abedon, S. T., and Loc-Carrillo, C. (2013). Phage cocktails and the future of phage therapy.
Expert Review of Anti-infective Therapy, 11(8),
755–760. https://doi.org/ 10.1586/14787210.2013.819725
Chaudhry, W.
N., Concepción-Acevedo, J., Park, T., Andleeb, S., Bull, J. J., and Levin, B.
R. (2017).
Synergy and order effects of antibiotics and phages in killing Pseudomonas aeruginosa biofilms. PLoS One, 12(1), e0168615.
https://doi.org/10.1371/journal.pone.
0168615
Clark, J. R., and March,
J. B. (2004). Bacteriophage-mediated nucleic
acid immunisation. FEMS
Immunology and Medical Microbiology, 40(1),
21–26. https://doi.org/10.1016/
S0928-8244(03)00301-6
Comeau, A. M., Tétart,
F., Trojet, S. N., Prère,
M. F., and Krisch, H. M. (2007).
Phage-antibiotic synergy (PAS): β-lactam
and quinolone antibiotics stimulate virulent phage
growth. PLoS One, 2(8), e799. https://doi.org/10.1371/journal.pone.0000799
Endersen, L.,
O’Mahony, J., Hill, C., Ross, R. P., McAuliffe, O., and Coffey, A. (2014).
Phage therapy in the food industry. Annual
Review of Food Science and Technology, 5,
327– 349. https://doi.org/10.1146/annurev-food-030713-092415
Fabijan, A. P., Lin, R. C. Y., Ho, J., Maddocks, S., Khatami, A., Lam, M., ... and Iredell, J. R. (2020).
Safety of bacteriophage therapy in severe Staphylococcus aureus infection. Nature Microbiology, 5(3), 465–472.
https://doi.org/10.1038/s41564-019-0634-z
Fischetti, V. A. (2005). Bacteriophage lytic enzymes: Novel anti-infectives. Trends in Microbiology, 13(10), 491–496. https://doi.org/10.1016/j.tim.2005.08.007
Fruciano, D. E., and Bourne, S. (2020). Phage therapy for animal health: Focus on aquaculture. Viruses, 13(1), 1. https://doi.org/10.3390/v13010001
Goodridge, L. D., and Abedon, S. T. (2003). Bacteriophage therapy for food safety: Challenges
and opportunities. Current Opinion in
Biotechnology, 14(3), 294–301. https:// doi.org/10.1016/s0958-1669(03)00062-9
Górski, A.,
Międzybrodzki, R., Węgrzyn, G., Jończyk-Matysiak, E., Borysowski, J., and Weber-Dąbrowska,
B. (2016). Phage therapy: Combating infections with potential for evolving from merely a last resort
to systematic application. Cellular
and Molecular Life Sciences, 73(23),
4473–4491. https://doi.org/10.1007/s00018-016-2286-7
Hatfull, G. F.,
Dedrick, R. M., and Schooley, R. T. (2022). Phage therapy for antibiotic- resistant bacterial
infections. Annual Review of Medicine, 73, 197–211.
https://doi.org/
10.1146/annurev-med-012221-083637
Hodyra-Stefaniak, K., Miernikiewicz, P., Drapała, J., Drab, M., Jonczyk-Matysiak, E., Lecion, D., ... and Górski, A. (2015). Mammalian
host-versus-phage immune response
determines phage fate in vivo. Scientific Reports, 5, 11174. https://doi.org/10.1038/ srep11174
Howard-Varona, C., Hargreaves, K. R., Abedon,
S. T., and Sullivan, M. B. (2017).
Lysogeny in nature: Mechanisms,
impact and ecology of temperate phages.
The ISME Journal, 11(7), 1511–1520. https://doi.org/10.1038/ismej.2017.16
Jault, P., Leclerc, T., Jennes, S., Pirnay, J. P., Que, Y. A., Resch, G., ... and PhagoBurn study group. (2019). Efficacy and tolerability of a
cocktail of bacteriophages to treat burn wounds infected
by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind
phase 1/2 trial. The Lancet Infectious
Diseases, 19(1), 35–45. https:// doi.org/10.1016/S1473-3099(18)30482-1
Jones, J. B., Jackson,
L. E., Balogh, B., Obradovic, A., Iriarte, F. B., and Momol, M. T. (2007).
Bacteriophages for plant disease
control. Annual Review of Phytopathology,
45, 245– 262. https://doi.org/10.1146/annurev.phyto.45.062806.094444
Jończyk-Matysiak, E., Weber-Dąbrowska, B., Owczarek, B., Międzybrodzki, R., Łusiak- Szelachowska, M., and Górski, A. (2021). Phage therapy in veterinary medicine: A historical and modern perspective. Antibiotics (Basel), 10(6),
680. https://doi.org/10.3390/
antibiotics10060680
Labrie, S. J., Samson,
J. E., and Moineau, S. (2010). Bacteriophage resistance mechanisms.
Nature Reviews
Microbiology, 8(5), 317–327. https://doi.org/10.1038/nrmicro2315
Lin, D. M., Koskella, B., and Lin, H. C. (2017). Phage therapy: An alternative to antibiotics in the
age of multi-drug resistance. World
Journal of Gastrointestinal Pharmacology and
Therapeutics, 8(3),
162–173. https://doi.org/10.4292/wjgpt.v8.i3.162
Loc-Carrillo, C., and Abedon, S. T. (2011). Pros and cons of phage therapy. Bacteriophage, 1(2), 111–114.
https://doi.org/10.4161/bact.1.2.14590
Malik, D. J.,
Sokolov, I. J., Vinner, G. K., Mancuso, F., Cinquerrui, S., Clokie, M. R. J.,
... and Garton, N. J. (2017).
Formulation, stabilisation and encapsulation of bacteriophage for phage therapy.
Advances in Colloid and Interface Science, 249, 100–133.
https://doi.org/ 10.1016/j.cis.2017.05.014
Merabishvili, M., Pirnay, J. P., Verbeken,
G., Chanishvili, N., Tediashvili, M., Lashkhi, N., ... and De
Vos, D. (2009). Quality-controlled small-scale production of a well-defined bacteriophage cocktail for use in human
clinical trials. PLoS One, 4(3), e4944. https:// doi.org/10.1371/journal.pone.0004944
Miedzybrodzki, R., Borysowski, J., Weber-Dabrowska, B., Fortuna, W., Letkiewicz, S., Szufnarowski,
K., Pawełczyk, Z., Rogóż, P., Kłak, M., Wojtasik, E., and Górski, A. (2012). Clinical
aspects of phage therapy. Advances in Virus Research, 83, 73–121. https:// doi.org/10.1016/B978-0-12-394438-2.00003-7
Nilsson, A. S. (2014).
Phage therapy--constraints and possibilities. Upsala Journal
of Medical Sciences, 119(2), 192–198. https://doi.org/10.3109/03009734.2014.902878
Pande, J., Szewczyk, M. M., and Grover, A. K. (2010). Phage display:
Concept, innovations, applications
and future. Biotechnology Advances, 28(6), 849–858. https://doi.org/ 10.1016/j.biotechadv.2010.07.004
Pelfrene, E., Willebrand, E., Cavaleiro Sanches,
A., Sebris, Z., and Cavaleri,
M. (2016). Bacteriophage
therapy: a regulatory perspective. Journal
of Antimicrobial Chemotherapy, 71(8), 2071–2074. https://doi.org/10.1093/jac/dkw157
Peng, H., and Chen, I. A. (2021). Phage-based diagnostic and therapeutic applications. International Journal of Molecular Sciences, 22(10),
5145. https://doi.org/10.3390/ ijms22105145
Pires, D. P., Cleto, S., Sillankorva, S., Azeredo, J., and Lu, T. K. (2016). Genetically engineered phages: A review of advances over the last decade. Microbiology and Molecular Biology Reviews, 80(3),
523–543. https://doi.org/10.1128/MMBR.00069-15
Pirnay, J. P.,
De Vos, D., Verbeken, G., Merabishvili, M., Chanishvili, N., Vaneechoutte, M., ... and Lavigne, R. (2011). The phage therapy paradigm: Prêt-à-porter or sur-mesure? Pharmaceutical Research, 28(4), 934–937.
https://doi.org/10.1007/s11095-010-0313-5
Principi, N., Silvestri, E., and Esposito, S. (2019). Phage therapy: An additional weapon
in the armamentarium against
multidrug-resistant bacterial infections? Frontiers
in Pharmacology, 10, 194. https://doi.org/10.3389/fphar.2019.00194
Puapermpoonsiri,
M., Spencer, J., and van der Walle, C. F. (2009). A freeze-dried formulation of bacteriophage K: An alternative approach to treating
Staphylococcus aureus infections. European Journal
of Pharmaceutics and Biopharmaceutics, 71(2), 222–229. https://doi.org/10.1016/j.ejpb.2008.09.011
Rhoads, D. D., Wolcott,
R. D., Kuskowski, M. A., Wolcott, B. M., Ward, L. S., and Sulakvelidze, A.(2009). Bacteriophage therapy of venous leg ulcers in humans: Results of a phase I safety trial. Journal of Wound Care, 18(6),
237–243. https://doi.org/10.12968/jowc. 2009.18.6.42801
Schooley, R. T., Biswas,
B., Gill, J. J., Hernandez-Morales, A., Lancaster, J., Lessor, L., Barr,
J. J., Reed, S. L., Rohwer, F., Benler, S., Segall, A. M., Taplitz,
R., Smith, D. M., Kerr,
K., Kumaraswamy, M., Nizet,
V., Lin, L., McCauley, M. D., Strathdee, S. A., Aslam, S., and Hamilton,
T. (2017). Development and use of personalized bacteriophage-based therapeutic cocktails to treat a patient
with a disseminated resistant Acinetobacter baumannii infection. Antimicrobial
Agents and Chemotherapy, 61(10),
e00954-17. https://doi.org/10.1128/AAC.00954-17
Segall, A. M., Roach, D. R., and Strathdee, S. A. (2019).
New frontiers: phage therapy against
bacterial pathogens of the opioid
epidemic. Drug and Alcohol Dependence,
198, 1–5. https://doi.org/10.1016/j.drugalcdep.2019.02.016
Smith, G. P. (1985). Filamentous fusion phage: Novel expression vectors that display cloned
antigens on the virion surface.
Science, 228(4705), 1315–1317. https://doi.org/
10.1126/science.4001944
Sulakvelidze, A., Alavidze, Z., and Morris, J. G., Jr. (2001).
Bacteriophage therapy. Antimicrobial Agents and Chemotherapy, 45(3),
649–659. https://doi.org/10.1128/AAC. 45.3.649-659.2001
Summers, W. C. (1999). Félix d’Herelle
and the origins of molecular
biology. Yale University Press.
Tagliaferri, T.
L., Compagnone, M., Sahi, J. S., Riello, G., Von Arx, C., and Ruga, S. (2021). Phage-antibiotic combination therapy: State of the art and future perspectives. Critical
Reviews in Microbiology, 47(6), 719–736.
https://doi.org/10.1080/1040841X.2021.1969869
Verbeken, G., Pirnay, J. P., De Vos, D., Jennes, S., Zizi, M., Lavigne, R., ... and Huys, I. (2014). Optimizing bacteriophage therapy: In vivo studies matter.
Expert Review of Anti-infective Therapy, 12(7), 753–757.
https://doi.org/10.1586/14787210.2014.915687
World Health Organization. (n.d.). Antibiotic
resistance. Retrieved June 2, 2025, from https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
Yacoby, I., Bar, H., and Benhar, I. (2007). Targeted cancer therapy by employing engineered bacteriophages. Proceedings of the National
Academy of Sciences
of the United States of America, 104(45), 17950–17955. https://doi.org/10.1073/pnas.0704112104
Yosef, I., Manor, M., Kiro, R., and Qimron, U. (2015). Temperate
and lytic bacteriophages programmed to sensitize bacteria to antibiotics. Proceedings of the National Academy of Sciences
of the United States of America, 112(14),
4266–4271. https://doi.org/10.1073/
pnas.1500107112
Young, R. (1992). Bacteriophage lysis: Mechanism and regulation. Microbiological Reviews, 56(3), 430–481.
https://doi.org/10.1128/mr.56.3.430-481.1992
Łusiak-Szelachowska, M., Żaczek, M., Weber-Dąbrowska, B., Międzybrodzki, R., Kłak, M., Fortuna, W., ... and Górski,
A. (2014). Phage neutralization by sera of patients receiving phage therapy. Viral Immunology, 27(6), 295–304.
https://doi.org/10.1089/vim.2013.0128