The Efficacy and Clinical Application of Mefenamic Acid in Odontogenic
Pain Management: A Comprehensive Review
*Zakir Hidayatallah
Division of Basic Medical Sciences, Department
of Basic and Clinical Oral Sciences, Faculty of Dental Medicine, Umm Al-Qura University,
Makkah, Saudi Arabia
*Corresponding author: Zakir Hidayatallah Email: Zhmarwat@uqu.edu.sa
Received: 25-03-2025, Accepted:
03-04-2025, Published
online: 22-04-2025
DOI: https://doi.org/10.33687/ricosbiol.03.04.0059
Abstract
Odontogenic
pain, arising from inflammation of dental and periodontal tissues, poses a
common and challenging issue in dental practice. It impacts patient comfort,
treatment adherence, and overall oral health. Effective pain management is
essential for achieving successful clinical outcomes. Mefenamic acid is widely
prescribed due to its efficacy in managing various dental pain conditions. This
review evaluates the effectiveness, safety, and mechanisms of action of
mefenamic acid in treating odontogenic pain, including primary pain,
post-extraction pain, pulpitis, and periodontal pain. A systematic review was
conducted on clinical studies, randomized controlled trials, and meta-analyses
sourced from the PubMed, Scopus, and Cochrane databases. The study aimed to
examine the analgesic effects, adverse effects, and other clinical applications
of mefenamic acid compared with other NSAIDs used in dentistry. Mefenamic acid
has demonstrated analgesic properties, likely resulting from its inhibition of
the cyclooxygenase (COX) enzymes, which decreases pain and the inflammatory
response associated with prostaglandin synthesis. Clinical research has
consistently validated its efficacy in treating post-extraction, pulpitis, and
periodontal pain. However, gastrointestinal side effects, particularly
dyspepsia and other forms of gastropathy, are the most documented issues,
necessitating careful case selection and follow-up. Research suggests that
mefenamic acid is at least as effective as some common non-steroidal
anti-inflammatory drugs, including ibuprofen and diclofenac, but is less
harmful to the stomach than ibuprofen. Mefenamic acid is recognized as a viable
pain management option in dentistry, but its clinical use requires further
study. Specifically, research should determine optimal dosage, treatment
duration, and effectiveness. This research should also compare mefenamic acid
to newer, more selective NSAIDs.
Furthermore,
ongoing research into novel drug design, pharmacogenetic factors influencing
NSAID efficacy, and alternative pain treatment methods is likely to enhance the
clinical utility of mefenamic acid. Mefenamic acid is an effective option for
managing odontogenic pain; however, careful patient selection and monitoring
are crucial to minimize potential adverse effects. Future research should
concentrate on optimizing its application.
Keywords:
Mefenamic acid, NSAIDs, odontogenic pain, pain management, COX inhibitors.
Introduction
Patients with dental
caries, periodontitis, or post-extraction complications commonly report odontogenic
pain (Pak & White, 2011; Fitriyati et al.., 2021).
Effective pain management is crucial
for successful clinical outcomes. Nonsteroidal anti-inflammatory drugs (NSAIDs)
exert therapeutic and toxic effects through a common mechanism of action: COX-1
inhibition. For pain and inflammation treatment after a dental surgical procedure,
short-term use of NSAIDs (typically one week or less) is effective and safe (Bushra
& Aslam, 2010). Compared with opioid combination drugs, NSAIDs do not have as
many unwanted CNS depressant effects that are responsible for the high rates of
drowsiness, dizziness, nausea, and constipation often encountered with opioid-containing
products (Elvir-Lazo & White, 2010).
The FDA now
grants over-the-counter (OTC) status to ibuprofen, naproxen, ketoprofen, and
mefenamic acid. Several NSAIDs, including these medications, are available OTC,
which increases their accessibility for managing dental pain (Bushra &
Aslam, 2010). However, adhering to the recommended dosages and durations
outlined in OTC regulations is crucial to minimize potential adverse effects.
OTC regulations specify that these medications must not be used for more than
10 consecutive days for pain and only 3 days for fever, along with daily and
single dose limits that are lower than the prescribed usage of the drugs.
The adverse
effects of prolonged NSAID treatment, particularly gastrointestinal ulcers,
perforations, and bleeding, appear to have been reduced by the introduction of
highly selective COX-2 inhibitors. Trials aimed at preventing large colon
polyps have shown an increased risk of cardiovascular events when these
medications were compared to a placebo. Even short-term use (10 days) of these
drugs for post-operative pain following coronary artery bypass graft surgery
has resulted in a significant increase in risk.
This has led to
the global withdrawal of most selective COX-2 inhibitors from the market (Dahl
& Møiniche, 2004; Al-Sukhun et al.., 2012).
NSAIDs are
among the most commonly used agents in dentistry, as they not only relieve pain
but also reduce inflammation. Mefenamic acid, which belongs to the fenamate
group of NSAIDs, is renowned for its potent analgesic and anti-inflammatory
properties. Patients with dental caries, periodontitis, or post-extraction
complications often report experiencing odontogenic pain (Pak & White,
2011; Fitriyati et al., 2021). This review evaluates the efficacy, safety, and
pharmacological profile of mefenamic acid in managing odontogenic pain. It
compares mefenamic acid to other NSAIDs and explores potential future
directions. Furthermore, this review synthesizes the first direct comparisons
between mefenamic acid and newer NSAIDs in treating dental pain while proposing
future pharmacogenetic and formulation-driven approaches.
Materials and Methods:
Search Strategy and Methodology
An assessment
was conducted using a systematic review approach to evaluate mefenamic acid's
clinical effectiveness, safety, and pharmacological actions in managing
odontogenic pain. The following electronic databases were searched: PubMed,
Scopus, and the Cochrane Library. The search strategy employed Medical Subject
Headings (MeSH) terms and keywords to identify all scientific works on the
issue under review (Higgins et al., 2019). The search was restricted to human
studies and articles published in English. Initially, no time limits were set
to encompass all relevant studies; however, this was later analyzed by focusing
on the most recent publications, such as those from the last decade, to discern
trends and evidence.
Selection Criteria for Studies
Studies were considered for analysis
if the criteria listed below were met:
- Clinical trials (randomized controlled and
controlled clinical trials) evaluating the effectiveness and safety of
mefenamic acid for managing odontogenic pain.
- Systematic reviews and meta-analyses that
focus on using mefenamic acid in dentistry.
- Investigations comparing mefenamic acid
with other analgesics or anti-inflammatory treatments in odontogenic pain
(Santini et al., 2021; Kumar, Sangwan, & Tewari, 2021).
- Most studies analysed short-term outcomes;
thus, conclusions about chronic use or rare adverse effects (e.g., SJS) remain
tentative (Santini et al., 2021).
The following criteria were used to rule
participants out of the study:
- Animal subjects: lab-based
experimentation.
- Non-English publications (credible
articles discovered through the search have been archived for potential
future development and analysis).
- Non-English case reports or case series
involving fewer than ten participants, as well as articles unrelated to
the treatment of odontogenic pain.
Data Extraction and Quality Assessment
Standardised data
extraction forms were utilised in the remaining studies to collect information.
The gathered information consisted of:
- Type of Research
- Characteristics of the patient population
studied (e.g., age, sex)
- Intervention(s): Administering mefenamic
acid (including dosage) and other comparison drugs for mefenamic acid
- Results (pain intensity, pain alleviation,
adverse effects)
- Follow-up Duration
The Cochrane
Risk of Bias tool was utilized to identify various sources of bias, such as
selection, performance, detection, attrition, and reporting (Higgins et al.,
2019). The overall quality of evidence for outcomes was evaluated using the
GRADE (Grading of Recommendations Assessment, Development, and Evaluation)
methodology (Guyatt et al., 2011).
Synthesis of the Data
The narrative
synthesis involved summarizing and explaining the findings of the included
studies in a descriptive format. This approach enabled the integration of data
from studies with varying designs and outcomes, offering a comprehensive
overview of mefenamic acid's effectiveness and safety. Some of the search terms
included the following: Mefenamic Acid, NSAIDs, odontogenic pain, dental pain,
post-extraction pain, pulpitis, periodontitis pain, analgesics, and
inflammatory mediators. Meta-analyses were conducted only when there was
sufficient evidence and it was deemed appropriate (Santini et al., 2021;
Guerreiro et al., 2021).
PRISMA SELECTION STUDY OVERVIEW 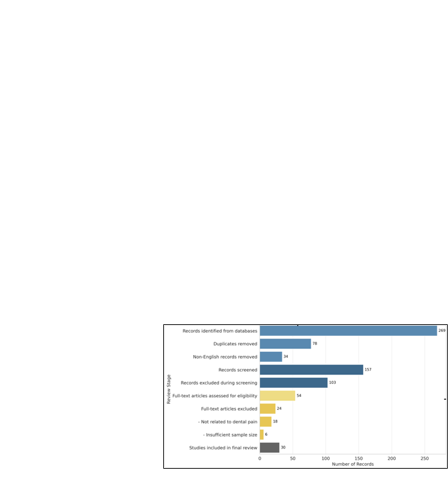
Pharmacological
Profile
Pharmacokinetics
Mefenamic acid
has a high oral bioavailability of approximately 90%, due to its lipophilic
characteristics. The peak plasma concentration of 10–20 µg/mL is reached 2–4
hours after administering a 500 mg dose. It undergoes extensive hepatic
metabolism, resulting in the formation of inactive glucuronide metabolites that
are excreted through the kidneys and bile. Other active glucuronide metabolites
have a half-life (T1/2) of 2–4 hours; thus, dosing should be conducted every 6
hours to maintain therapeutic analgesic levels. Food Interaction: Food does not
significantly influence mefenamic acid, as it may delay absorption without
considerably affecting overall bioavailability. This is advantageous for
patients who need to take the medication with food to reduce the risk of
gastrointestinal irritation (Bushra & Aslam, 2010).
Pharmacodynamics
Mefenamic acid is an anti-inflammatory analgesic that inhibits
cyclooxygenase (COX) enzymes responsible for synthesizing prostaglandins (PGs).
These chemical substances play a role in pain, inflammation, and fever; hence,
their inhibition is significant (Kelly, Ahmad, & Brull, 2001).
·
COX-1 Inhibition: Mefenamic acid inhibits COX-1, which is present
in most tissues and is crucial for maintaining normal physiological functions,
such as protecting the gastric mucosa and regulating renal blood flow. The
inhibition of COX-1 is associated with adverse gastrointestinal effects,
including gastric irritation and ulceration (Dahl & Moiniche, 2004).
·
COX-2 Inhibition: Furthermore, mefenamic acid inhibits COX-2,
located at inflammation sites and contributes to pain and swelling. By blocking
COX-2, mefenamic acid reduces the production of pro-inflammatory
prostaglandins, thus providing pain relief without disrupting normal bodily
processes (Albuquerque et al., 2017).
Many
nonselective COX-inhibiting NSAIDs, including mefenamic acid, tend to have more
significant adverse effects because they are stronger or at least equally
strong COX-1 antagonists. Unlike selective COX-2 inhibitors (e.g., celecoxib),
mefenamic acid possesses
Table 1: Comprehensive
Pharmacokinetic Parameters of Mefenamic Acid
|
Parameter |
Description |
Value/Range |
Clinical Implications |
References |
|
Oral Bioavailability |
Fraction absorbed
into systemic circulation |
~90% |
High absorption
ensures predictable dosing |
Bushra & Aslam,
2010 |
|
T<sub>max</sub> |
Time to peak plasma
concentration |
2–4 hours |
Take 1–2 hours
before painful procedures |
Team Medical Mini-Note,
2017 |
|
C<sub>max</sub> |
Peak plasma concentration
(500 mg dose) |
10–20 µg/mL |
Correlates with
analgesic efficacy |
Bushra & Aslam,
2010 |
|
Half-life (t<sub>½</sub>) |
Plasma elimination
half-life |
2–4 hours |
Requires q6h dosing
for sustained relief |
FDA Label, 2020 |
|
Protein Binding |
Fraction bound
to plasma proteins |
>90% (mainly
albumin) |
Caution in hypoalbuminemia
(↑ free drug levels) |
Albuquerque et
al., 2017 |
|
Metabolism |
Primary pathways |
CYP2C9 (80%) →
Glucuronidation |
CYP2C9 poor metabolizers
need dose reduction |
Guyatt et al.,
2011 |
|
Active Metabolites |
Hydroxymefenamic
acid glucuronide |
Yes (weak activity) |
Prolonged effect
in renal impairment |
Usman et al.,
2012 |
|
Excretion |
Primary route |
Urine (60%), Feces
(40%) |
Avoid in severe
renal/hepatic dysfunction |
Bushra & Aslam,
2010 |
|
Food Effects |
Impact of food
on absorption |
Delays T<sub>max</sub>
by ~1h |
No dose adjustment
needed; take with food for GI protection |
Team Medical Mini-Note,
2017 |
Anti-inflammatory properties arise
because it inhibits both COX-1 and COX-2; however, this also increases the risk
of gastrointestinal damage. There is little debate that the series of reactions
in many painful conditions involves prostaglandins (PGs) and that aspirin’s
effectiveness in managing inflammation and pain is closely linked to its
inhibition of the inflammatory response and PG synthesis (Bushra & Aslam,
2010). Microdialysis techniques have demonstrated that following dental
surgery, the analgesic effects of NSAIDs correlate with a reduction in local PG
levels. However, some evidence suggests that the analgesic and
anti-inflammatory actions of NSAIDs may occur through different mechanisms
(Dahl & Møiniche, 2004). Firstly, there is a difference in the timing of
onset between the analgesic and anti-inflammatory effects. Generally,
significant analgesia is achieved within one hour of drug administration. In
contrast, the inflammatory effect can take several days or weeks to reach its
peak levels due to the chronic nature of inflammatory processes. Additionally,
the maximum analgesic effect in humans is usually attained at lower doses than
those needed for antirheumatic and other anti-inflammatory effects (Kelly et
al., 2001).
- Other Mechanisms: Ion Channels and Neurogenic
Inflammation
Beyond COX inhibition,
emerging evidence suggests mefenamic acid may also modulate pain through additional
pathways:
Modulation of Voltage-Gated
Sodium Channel Activity (Nav1.7, Nav1.8): Mefenamic acid has been shown to decrease
the excitability of pain-transmitting sodium channels, which diminishes nerve activity
in inflammatory states (Bushra & Aslam, 2010).
Inhibition of Nuclear Factor Kappa B
(NF-κB) Signalling: Mefenamic acid inhibits the production of pro-inflammatory cytokines,
such as IL-6 and TNF-α. Unrestricted use of mefenamic acid increases the amount
of anti-inflammatory and analgesic cytokines. Thus, the cytokines responsible for
inflammation are blocked together with the pathways responsible for their generation
(Kelly et al., 2001).
The additional mechanisms further enhance
the already broad-spectrum effectiveness of the drug in inflammatory pain, such
as odontogenic pain. However, these mechanisms are still under active research,
and clinical translation of this evidence remains ongoing.
Clinical Applications
Mefenamic acid has demonstrated effectiveness in managing various forms
of dental pain.
·
Post-Extraction Pain: Mefenamic acid is as effective as ibuprofen (Bailey et al.,
2013).
· Pulpitis: Validated efficacy in clinical studies (Pangalila et al., 2016; Kumar et al., 2021).
·
Periodontal
Pain: Mefenamic acid has also been shown to be effective
in treating periodontal pain (Bailey et al.,
2013; Santini et al., 2021).
Comparative Efficacy
with Other Analgesics
The effectiveness
of mefenamic acid in controlling odontogenic pain has been demonstrated across multiple
study designs, though variations in populations and outcome measures require careful
interpretation (Santini et al., 2021; Kumar et al., 2021; Bailey et al.,
2013). Comparative trials consistently show the relationship between these variables.
Mefenamic Acid vs Ibuprofen
Clinical evidence
confirms that mefenamic acid has comparable efficacy to ibuprofen for post-extraction
pain (Bailey et al., 2013) and pulpitis (Pangalila et al., 2016; Bushra
& Aslam, 2010). Some studies suggest mefenamic acid may offer better
tolerability of gastrointestinal (Santini et al., 2021). This difference
may be clinically significant for gastrointestinal intolerance, a common reason
for NSAID discontinuation. It is also important to note that, in practice, the extent
of this difference is likely to vary between studies and should be considered along
with other patient-related variables. For example, people with a previous history
of gastric ulcers may be better off with mefenamic acid, while those for whom ibuprofen
worked may continue with it.
- Mefenamic
Acid vs Diclofenac/Naproxen
Meta-analyses suggest
that mefenamic acid, naproxen, and diclofenac have similar effectiveness in managing
pain associated with dental procedures (Pangalila et al., 2016; Santini et
al., 2021). In clinical trials, mefenamic acid demonstrated a reduced
incidence of adverse effects relative to naproxen and diclofenac (Pangalila et
al., 2016). In cases of acute inflammation, diclofenac proved to be more effective
than the others, highlighting the importance of considering the type of odontogenic
pain being treated. When inflammation is a significant symptom, the drug that
is used may be diclofenac. However, for patients with gastrointestinal complications,
mefenamic acid is suggested. The degree of drug response is unique to each patient,
and factors such as age, comorbidities, and concomitant medications must be considered
(Bushra & Aslam, 2010).
- Mefenamic
Acid vs. Selective COX-2 Inhibitors
Mefenamic acid is not an exception to
the general rule regarding its effect on the GI mucosa; certain risks to the functioning
of the gastrointestinal tract accompany its use. On the other hand, mefenamic acid
is more advantageous in providing rapid pain control than selective COX-2 inhibitors,
such as celecoxib, which have a better gastrointestinal side effect profile (Al-Sukhun
et al., 2012). Mefenamic acid may provide more rapid analgesia and be beneficial
in post-operative inflammation and more severe cases. Where there is a need for
chronic pain management, mefenamic acid may not be as helpful due to the constant
requirement of long-term NSAID use. However, it is suggested for acute dental pain,
where a more rapid analgesic response is desired. There are concerns of cardiovascular
side effects with COX-2 inhibitors, which must be considered in comparison to the
gastrointestinal side effects of non-selective NSAIDs (Dahl & Moiniche, 2004).
Safety Profile and
Adverse Effects
When considering
the use of mefenamic acid (NSAIDs), particular attention must be paid to its anticipated
adverse effects: gastrointestinal, cardiovascular, renal, CNS, and dermatological
reactions. Such risks are critical for the proper clinical application (Bushra &
Aslam, 2010; Handisurya et al).
Figure 1: Comparative Pain Relief Profiles of NSAIDs
Source: Adapted from Bailey et al., (2013) and Pangalila et al. (2016).
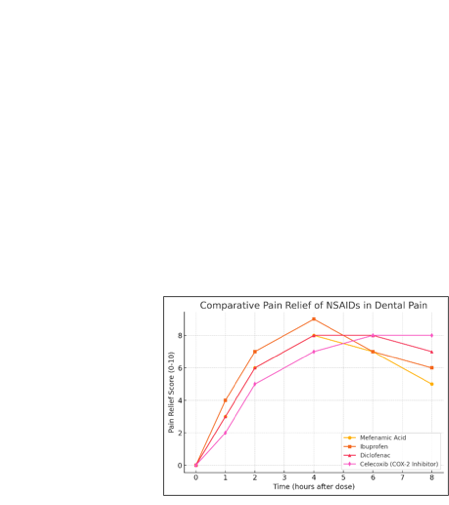
Figure 1: Ibuprofen provides
the highest pain relief but has a higher risk of gastric side effects.
·
Mefenamic acid provides comparable pain relief to diclofenac but may
offer a better safety profile.
·
Celecoxib, a COX-2 selective inhibitor, provides long-lasting relief
but has a delayed onset.
- Gastrointestinal Risks
Mefenamic acid may induce adverse effects, including gastric irritation
and ulceration (Handisurya et al., 2011; Bailey et al., 2013). The
risk is elevated in patients with a history of peptic ulcer disease, though concomitant
Proton Pump inhibitor use may mitigate this (Guyatt et al., 2011). Protective strategies do not eliminate
the risk of GI symptoms entirely
- Cardiovascular Concerns
NSAIDs, including
mefenamic acid, can elevate blood pressure and increase cardiovascular risks (myocardial
infarction, stroke), particularly with prolonged use (Dahl & Møiniche, 2004;
Al-Sukhun et al., 2012). These effects are dose- and duration-dependent.
Patients with pre-existing cardiovascular
conditions should use mefenamic acid cautiously, at the lowest effective dose, and
for the shortest duration possible.
- Renal Toxicity
Mefenamic acid may
impair renal blood flow, leading to fluid retention and elevated creatinine, especially
in patients with chronic kidney disease or dehydration (Bushra & Aslam, 2010).
Short-term use (<7 days) is preferred in at-risk populations (Bailey et al.,
2013).
Comorbid conditions such as congestive
heart failure or dehydration can further exacerbate renal risk.
- CNS
Toxicity
Mefenamic acid may
lower seizure thresholds, particularly in adolescents or overdose scenarios (Doğan
et al., 2019). While preclinical studies suggest potential anticonvulsant
properties, clinical evidence remains cautionary.
- Skin
Allergies
Mefenamic acid also
can cause skin allergies, including fixed drug eruptions (FDE), urticaria, and rare
but severe reactions like Stevens-Johnson syndrome (SJS). Case reports and pharmacovigilance
data highlight these adverse effects, with some studies ranking it among NSAIDs
linked to cutaneous reactions (Handisurya A et al., 2011).
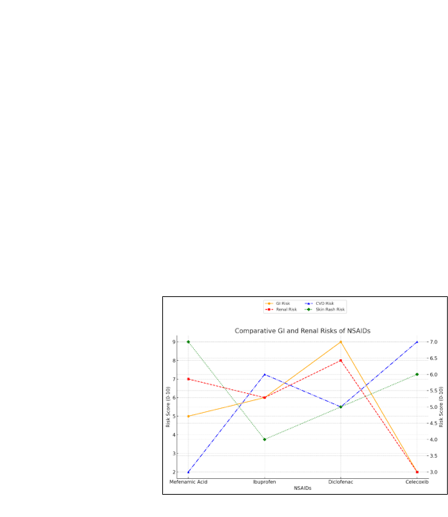
Figure 2: Comparative GI/Renal/CVD/Derma Risks of NSAIDs
(Sources: Bushra & Aslam, 2010; Dahl & Møiniche, 2004; Handisurya et
al., 2011; Al-Sukhun et al., 2012)
Clinical Guidelines and Patient-Specific Recommendations
Dosage and Administration
Mefenamic acid is typically prescribed for short-term use (3–7 days)
to optimize pain relief while minimizing adverse effects. This duration aligns with
its pharmacokinetic profile (peak plasma concentration Cmax = 10–20 µg/mL)
and gastrointestinal tolerability (Bailey et al., 2013; Team Medical Mini-Note,
2017).
Emerging Research and Future Directions
The study and management of odontogenic
pain remains a progressing field with new research aimed at optimizing analgesic
therapies and their side effects. A few possibilities are most noteworthy and require
further research to increase the clinical effectiveness of mefenamic acid and improve
patient outcomes.
Table 2: Dosage
and Administration
|
Patient Group |
Dosage |
Frequency |
Max Duration |
Key References |
|
Adults |
500 mg initial
dose → 250 mg maintenance |
Every 6 hours |
7 days |
Bailey et al.,
2013 |
|
Elderly (>65
yrs) |
250 mg |
Every 8 hours |
5 days |
Team Medical Mini-Note,
2017 |
|
Children (>12
yrs) |
250 mg |
Every 6–8 hours |
5 days |
Bushra & Aslam,
2010; Team Medical Mini-Note, 2017 |
Table 3: Indications
& Contraindications of Mefenamic Acid in Dental Practice
|
Category |
Clinical Scenario |
Rationale |
Key References |
|
INDICATIONS |
|||
|
Post-extraction
pain |
Pain after tooth
removal |
Comparable efficacy
to ibuprofen, with faster onset (Pangalila et al., 2016; Bailey et al.,
2013). |
Bailey et al.,
2013; Pangalila et al., 2016 |
|
Acute pulpitis |
Inflamed dental
pulp pain |
Effective prostaglandin
inhibition at pain source (Pak & White, 2011; Kumar et al., 2021). |
Pak & White,
2011; Kumar et al., 2021 |
|
Periodontal pain |
Gingivitis/periodontitis
pain |
Reduces inflammation
in soft tissues (Santini et al., 2021). |
Santini et
al., 2021 |
|
Post-surgical
pain |
Post-oral surgery
discomfort |
Short-term use
aligns with COX-2 inhibition (Al-Sukhun et al., 2012). |
Al-Sukhun et
al., 2012 |
Table 4: Contraindications
of Mefenamic Acid in Dental Practice
|
Contraindication |
Risk Level |
Mechanism |
Clinical Rationale |
Alternatives |
Key References |
|
Active peptic
ulcer |
High (Absolute) |
COX-1 inhibition
→ Reduced gastric cytoprotection |
5x higher GI bleeding
risk vs. non-NSAID users; avoid even with PPI co-therapy |
Acetaminophen,
celecoxib + PPI |
Handisurya et
al., 2011 |
|
Severe renal impairment
(eGFR <30) |
High |
Prostaglandin-dependent
renal vasoconstriction |
NSAIDs precipitate
AKI in 18-25% of CKD patients |
Opioids (short-term) |
Bushra & Aslam,
2010 |
|
Post-MI/CABG (<6
months) |
High |
COX-2 inhibition
→ Thrombosis risk |
2.5x increased
CV event recurrence; avoid all NSAIDs |
Non-NSAID analgesics |
Dahl & Møiniche,
2004 |
|
Children (<12
years) |
Moderate-High |
CNS penetration
→ Seizure threshold lowering |
Case reports of
seizures in adolescents; no FDA approval for dental pain <14y |
Ibuprofen (weight-based) |
Doğan et al.,
2019 |
|
3rd trimester
pregnancy |
Absolute |
Premature ductus
arteriosus closure |
Fetal renal toxicity
risk; contraindicated after 30 weeks |
Acetaminophen |
FDA Drug Safety
Communication, 2020 |
|
NSAID/aspirin
allergy |
Variable |
Cross-reactive
hypersensitivity |
30% risk of cross-reactivity
with other NSAIDs (esp. urticaria/FDE) |
Opioid alternatives |
Handisurya et
al., 2011 |
- New
Formulations
Several attempts are being made to modify
the dosage forms of mefenamic acid, with the aim of improving its pharmacokinetic
profile and reducing systemic side effects. For example, nanoparticle formulations
have been demonstrated to enhance drug solubility and bioavailability, as well as
mitigate off-target therapeutic effects, by delivering drugs to specific sites (Sriamomsak
et al., 2015; Shah, Shrivastava, & Mishra, 2013). Another transdermal
route of drug delivery currently being studied is the use of transdermal patches,
which may aid in treating certain types of odontogenic pain, such as temporomandibular
joint pain or postoperative pain relief following minor oral surgery. Sustained-release
oral formulations are also being developed to provide longer-lasting pain relief,
reduce dosing frequency, and improve adherence while maintaining consistent plasma
drug concentrations (Khullar et al., 2012; Usman et al., 2012). These
new formulations require additional clinical trials to evaluate their effectiveness
and safety in various patient populations.

Figure 3: The conceptual framework derived
from Sriamomsak et al., (2015) and Usman et al., (2012).
- Pharmacogenetics
The study of pharmacogenetics—which is
known to predict drug response—appears particularly helpful for customizing pain
management. Polymorphisms in the CYP2C9 gene influence the metabolism of mefenamic
acid, potentially impacting its pharmacokinetics, potency, and the risk of side
effects. Recognizing patients with specific CYP2C9 genotypes could help clinicians
adjust mefenamic acid dosing to maximize therapeutic effects and minimize adverse
outcomes (Guyatt et al., 2011). Future studies should confirm these pharmacogenetic
associations in larger, more diverse patient populations and support the development
of clinically actionable genotyping tools. Pharmacogenetic
screening (e.g., CYP2C9 polymorphisms) could personalize mefenamic acid dosing,
while transdermal patches may benefit patients with TMJ pain who are at risk of
gastrointestinal events.
- Other Ways of Managing Pain
In addition to pharmaceutical advances,
adjunctive or alternative therapies are increasingly being considered in the management
of odontogenic pain. Combination therapies—such as locally administered NSAIDs with
local anesthetics—may improve efficacy while reducing systemic exposure and side
effects (Penprase et al., 2015). Other non-traditional approaches, including
lasers, acupuncture, or cognitive behavioral therapy, are being evaluated for their
potential to manage chronic dental pain and complement standard analgesics (Guerreiro
et al., 2021). More research is required to identify optimal combinations
of pharmacologic and non-pharmacologic strategies tailored to specific types of
odontogenic pain and individual patient profiles.
Clinical Guidelines
and Recommendations
·
Mefenamic acid shows comparable efficacy to ibuprofen in managing
odontogenic pain, with a slightly better gastrointestinal safety profile
(Bushra & Aslam, 2010; Bailey et al., 2013).
·
However, thorough patient selection and monitoring are crucial to
reduce potential adverse effects.
·
Patients with a history of gastric ulcers may benefit more from
mefenamic acid, while those for whom ibuprofen was effective can continue using
it.
·
When inflammation is a major symptom, the medication of choice may
be diclofenac.
Conclusions
Future research should prioritize:
·
Optimizing drug delivery while minimizing systemic side effects
through novel formulations (Sriamomsak et al., 2015; Usman et al., 2012).
·
Develop pharmacogenetic strategies to personalize mefenamic acid dosing
(Guyatt et al., 2011).
·
Evaluating new analgesics and combination therapies for various
odontogenic pain conditions (Santini et al., 2021; Guerreiro et al., 2021).
·
Integrating non-pharmacological approaches to enhance systemic
pharmacological treatments (Penprase et al., 2015).
These advancements, along with comprehensive education and training
for dental practitioners, will enhance the management of odontogenic pain and
improve patient outcomes.
References:
Albuquerque, A. F. M., Fonteles, C. S.
R., do Val, D. R., et al. 2017. Effect of Preemptive Analgesia on Clinical
Parameters and Tissue Levels of TNF-α and IL-1β in Third Molar Surgery: A Triple-Blind,
Randomized, Placebo-Controlled Study. Int J Oral Maxillofac Surg 46: 1615-1625.
Al-Sukhun, J., Al-Sukhun, S., Penttilä,
H., Ashammakhi, N., and Al-Sukhun, R. 2012. "Preemptive Analgesic Effect of
Low Doses of Celecoxib is Superior to Low Doses of Traditional Nonsteroidal Anti-inflammatory
Drugs." J Craniofac Surg 23: 526-529.
Bailey, E., Worthington, H. V., van Wijk, A., Yates, J. M., Coulthard,
P., and Afzal, Z. 2013. "Ibuprofen and/or Paracetamol (Acetaminophen) for Pain
Relief After Surgical Removal of Lower Wisdom Teeth." Cochrane Database Syst
Rev.
https://doi.org/10.1002/14651858.CD004624.pub2.
Bauer, H. C., Duarte, F. L., Horliana,
A. C. R. T., et al. 2013. "Assessment of Preemptive Analgesia with Ibuprofen
Coadministered or Not with Dexamethasone in Third Molar Surgery: A Randomized Double-blind
Controlled Clinical Trial." Oral Maxillofac Surg 17: 165-171.
Bushra, R., and Aslam, N. 2010. "An
Overview of Clinical Pharmacology of Ibuprofen." Oman Med J 25: 155-1661.
Chagas Carvalho Alves, N., Raiane Mamede
Veloso, S., de Andrade Silva, S., et al. 2021. "Influence of Occlusal
Reduction on Pain After Endodontic Treatment: A Systematic Review and Meta-Analysis."
Sci Rep. https://doi.org/10.1038/s41598-021-93119-6.
Chiu, W. K., and Cheung, L. K. 2005. "Efficacy
of Preoperative Oral Rofecoxib in Pain Control for Third Molar Surgery." Oral
Surg Oral Med Oral Pathol Oral Radiol Endod. https://doi.org/10.1016/j.tripleo.2005.02.075.
Comoglo, T., Gonul, N., and Baykara,
T. 2003. "Preparation and In Vitro Evaluation of Modified Release Ketoprofen
Microsponges." Farmaco 58(2): 101-106.
Coulthard, P., Haywood, D., Tai, M. A.,
Jackson-Leech, D., Pleuvry, B. J., and Macfarlane, T. V. 2000. "Treatment of
Postoperative Pain in Oral and Maxillofacial Surgery." Br J Oral Maxillofac
Surg 38: 588-592.
Dahl, J. B., and Møiniche, S. 2004. "Pre-emptive
Analgesia." Br Med Bull 71: 13-27.
Elvir-Lazo, O. L., and White, P. F. 2010.
"The Role of Multimodal Analgesia in Pain Management After Ambulatory Surgery."
Curr Opin Anaesthesiol 23: 697-703.
Fadhilah, R. N. 2016. "Study of Drug Use in
Osteoarthritis Patients." Surabaya: Airlangga University, Department of Clinical
Pharmacy.
Fitriyati, Her Nurcahyo, and Susiyarti. 2021. "Overview
of Prescribing Analgesic Drugs for Toothache Patients at the Pradipta Pharmacy."
Pharmaceutical Scientific Journal, Vol. x.
Goddard, G., Karibe, H., and McNeill, C. 2004.
"Reproducibility of Visual Analogue Scale (VAS) Pain Scores to Mechanical Pressure."
Cranio 22: 250-256.
Guerrero, M. Y. R., Monteiro, L. P. B., de Castro,
R. F., Magno, M. B., Maia, L. C., and da Silva Brandão, J. M. 2021. "Effect
of Low-Level Laser Therapy on Postoperative Endodontic Pain: An Updated Systematic
Review." Complement Ther Med. https://doi.org/10.1016/j.ctim.2020.102638.
Guyatt, G., Oxman, A. D., Akl, E. A., et al.
2011. "GRADE Guidelines: 1. Introduction - GRADE Evidence Profiles and Summary
of Findings Tables." J Clin Epidemiol 64: 383-394.
Handisurya, A., Moritz, K. B., Riedl,
E., Reinisch, C., Stingl, G., Wöhrl, S. J. 2011. "Cutaneous Sensitization to
Mefenamic Acid: A Case Report." J Dtsch Dermatol Ges. 9(5): 374-378. https://doi.org/10.1111/j.1610-0387.2011.07621.x.
Higgins, J. P. T., Thomas, J., Chandler,
J., et al. 2019. Cochrane Handbook for Systematic Reviews of Interventions.
2nd ed. Chichester: John Wiley & Sons.
Istiqomah. 2022. "Journal of Pharmacy and
Pharmaceutical Management (JFMK)." E-ISSN: 2829-2812, Vol. 1, No. 2, July-December,
60-65.
Jain, A., Deveda, P., Vyas, N., Chauhan,
J., Khambete, H., and Jain, S. 2011. "Development of Antifungal Emulsion-Based
Gel for Topical Fungal Infection(s)." International Journal of Pharmaceutical
Research and Development 2(12): 1825.
Khairunnisa. 2017. "Level of Patient
Knowledge and Rationality of Self-Medication in Three Pharmacies in Panyabungan
City."
Khullar, R., Kumar, D., Seth, N., Saini,
S. 2012. "Formulation and Evaluation of Mefenamic Acid Emulgel for Topical
Delivery." Saudi Pharmaceutical Journal 20(1): 63-67.
Kumar, G., Sangwan, P., and Tewari, S.
2021. "Effect of Premedication on Postoperative Pain After Root Canal Therapy
in Patients with Irreversible Pulpitis: A Systematic Review and Meta-Analysis."
J Dent Anesth Pain Med 21: 397-411.
Kumar, L., and Verma, R. 2010. "In
Vitro Evaluation of Topical Gel Prepared Using Natural Polymer." International
Journal of Drug Delivery 2: 58-63.
McArdle, L. W., and Renton, T. 2012.
"The Effects of NICE Guidelines on the Management of Third Molar Teeth."
Br Dent J 213: E8.
Morse, Z., Tump, A., and Kevelham, E.
2006. "Ibuprofen as a Pre-emptive Analgesic is as Effective as Rofecoxib for
Mandibular Third Molar Surgery." Odontology 94: 59-63.
Nurettin Özgür Doğan, Sevinç Taş Çaylak,
and Serkan Yılmaz. 2019. "Central Nervous System Toxicity Due to Mefenamic
Acid." The American Journal of Emergency Medicine 37(3): 561.e1-561.e2. https://doi.org/10.1016/j.ajem.2018.11.027.
Pak, J. G., and White, S. N. 2011. "Pain
Prevalence and Severity Before, During, and After Root Canal Treatment: A Systematic
Review." J Endod 37: 429-438.
Pangalila, K., Wowor, P. M., and Hutagalung,
B. S. P. 2016. "Comparison of the Effectiveness of Giving Mefenamic Acid and
Diclofenac Sodium Before Tooth Extraction on the Duration of Pain Threshold After
Tooth Extraction." J e-GiGi 4(2): 124-130.
Penprase, B., Brunetto, E., Dahmani,
E., Forthoffer, J. J., and Kapoor, S. 2015. "The Efficacy of Preemptive Analgesia
for Postoperative Pain Control: A Systematic Review of the Literature." AORN
J 101: 94-105.
Rahbani Nobar, B., Dianat, O., Rahbani
Nobar, B., et al. 2021. "Effect of Rotary and Reciprocating Instrumentation
Motions on Postoperative Pain Incidence in Non-Surgical Endodontic Treatments: A
Systematic Review and Meta-Analysis." Eur Endod J 6: 3-14.
Sathorn, C., Parashos, P., and Messer,
H. 2008. "The Prevalence of Postoperative Pain and Flare-up in Single-and Multiple-visit
Endodontic Treatment: A Systematic Review." Int Endod J 41: 91-99.
Shamszadeh, S., Asgary, S., Shirvani,
A., Eghbal, M. J. 2021. "Effects of Antibiotic Administration on Postoperative
Endodontic Symptoms in Patients with Pulpal Necrosis: A Systematic Review and Meta-Analysis."
J Oral Rehabil 48: 332-342.
Shah, K., Shrivastava, S. K., and Mishra,
P. 2013. "Evaluation of Mefenamic Acid Mutual Prodrugs." Medicinal Chemical
Research 22(1): 70-77.
Singla, N., Hunsinger, M., Chang, P.
D., et al. 2015. "Assay Sensitivity of Pain Intensity Versus Pain Relief
in Acute Pain Clinical Trials: ACTTION Systematic Review and Meta-Analysis."
J Pain 16: 683-691.
Sriamomsak, P., Limmatvapirat, S., Piriyaprasarth,
S., Mansukmanee, P., and Huang, Z. 2015. "A New Emulsifying Formulation of
Mefenamic Acid with Enhanced Drug Dissolution." Asian Journal of Pharmaceutical
Sciences 10(2): 121-127.
Struzycka, I. 2014. "The Oral Microbiome
in Dental Caries." Polish Journal of Microbiology 63(2): 127–135. https://doi.org/10.33073/pjm-2014-018.
Swetha, A., Rao, G. M., Ramana, V. K.,
Niyaz, B. B., and Reddy, K. V. 2011. "Formulation and In Vitro Evaluation of
Etodolac Entrapped in Microsponge-Based Drug Delivery System." International
Journal of Pharmacy 1(2): 73-80.
Team Medical Mini-Note. 2017. Basic Pharmacology
& Drug Notes. MMN Publishing, Makassar. Directorate General of Pharmaceutical
Development and Health Devices. 2011.
Usman, M., Ali, I., Bibi, H., Iqbal, J., and Iqbal,
K. 2012. "Preparation and Evaluation of Controlled Release Tablets Containing
Mefenamic Acid." Journal of Clinical and Experimental Pharmacology 2(1): 107.
White, I. R., Bamias, C., Hardy, P., Pocock, S.,
and Warner, J. 2001. "Randomized Clinical Trials with Added Rescue Medication:
Some Approaches to Their Analysis and Interpretation." Stat Med 20: 2995-3008.
Zahra, A., and G. Zarei. 2016. "The Application
of Mefenamic Acid in Pain Management." Medical Journal of Pain 2(1): 15-21.