Physiological Influence of Licorice
Extract on Some Hormonal and Biochemical Parameters Alterations Induced by
Glucocorticoid in Male Rats
Safinaz Badie1 and Sohier Fathey2*
1Department of
Physiology, Al-Dayer, Jazan
University, Jazan 82912, Kingdom of Saudi Arabia.
2Department of Microbiology
and Immunology, National Research Centre, Dokki,
Egypt.
*Corresponding
author: Sohier Fathey Email: sohir_syam@yahoo.com
Received: 10-01-2025, Accepted: 05-02-2025, Published online:
20-02-2025
DOI: https://doi.org/10.33687/mtycxb04
Abstract
The current study investigates the
influence of licorice extract versus adrenal incapability induced by glucocorticoids
in male rats. Forty male rats were sectioned randomly into 4 groups. The first
was negative control group (G1): normal saline was given orally to rats. The
second was positive control group (G2): intra peritoneal treatment with
Hydrocortisone Sodium (50 mg/kg) for seven days. The third was therapeutic
group (G3): intra peritoneal treatment with Hydrocortisone Sodium (50 mg/kg)
for seven days and then licorice extract (100 mg /kg) given orally for 14 days.
The fourth was licorice extract Group (G 4): Rats were given 100 mg/kg of
licorice extract orally for 21 days. At the end of the experiment, hormonal
measurement; adrenocorticotropic hormone (ACTH), corticotropin-releasing
hormone (CRH), serum cortisol, as well as malondialdehyde (MDA), 11 β
-hydroxysteroid dehydrogenase enzyme (11β-HSD), in addition to serum sodium and
potassium were measured. The results demonstrated treatment with licorice
extract improved significantly (P<0.05) in ACTH, CRH and serum cortisol
hormones with non-significant reduction in serum MDA level in the therapeutic
group compared with the positive control group. Our results concluded that
licorice extract improves the alteration induced by hydrocortisone hormones and
reduces the free radicals.
Keywords | Adrenal incapability, Licorice
extract, 11β-HSD, Rats
Introduction
Adrenal incapability (AI) is a clinical disturbance that leads to
failure of the adrenal cortex to secrete or output cortisol. There are three
pathological types of adrenal incapability may present; primary, secondary and
tertiary. Particularly, primary adrenal incapability (PAI) which originated
from 2ry pathology of the adrenal gland which induces a defect at the adrenal
level is also accompanied by mineralocorticoid (aldosterone) deficiency (Fredrick
et al., 2024; Lewis et al., 2023;).
Synthetic glucocorticoids (GCs) are able to mitigate inflammation
and suppress the immune system, so they are frequently used as therapeutic
agents. Their likely side effect is
prohibition of the hypothalamus pituitary adrenal axis leading to adrenal
incapability (Díaz-Castro et al., 2020).
Various factors are elevating the prevalence of this type of
adrenal incapability such as the dose, route of administration, the duration of
therapy, and potency of glucocorticoid, in addition to individual sensitivity
and synchronized medicines that conflict with glucocorticoid metabolism. When
the therapy of the exogenous glucocorticoid medication is minimized, patients
may suffer signs of Cushing’s as well as glucocorticoid withdrawal syndromes.
So, prior to the return of adrenal function, the employ of glucocorticoids
shouldn’t be entirely cutout (Nachawi et al., 2024).
While extended utilization of exogenous glucocorticoids can induce atrophy of
adrenocortical layers and pituitary corticotroph
cells. Whereas, mineralocorticoids are regulated by the renin-angiotensin
system secreted to sustain aldosterone production, which (Borresen
et al., 2022).
Natural extract from the roots of licorice plants (Glycyrrhiza glabra), which is thought to contain active
components as glycyrrhizic acid which itself is
hardly absorbed from the alimentary tract. Prior to absorption, glycyrrhizin
acid is hydrolyzed to obtain glycyrrhetic acid, which
is the definitive biologically active metabolite (Yaw et al., 2015). Researchers have discussed the
beneficial uses of licorice extract at low accurate doses; anti-diabetic,
anti-hyperlipidemia, antioxidant, anti-inflammatory, antiviral, antimicrobial,
and anti-tumor merits. Moreover, it also has hepato-protective, neuro-protective
effects, renal protective, as well as thrombin inhibitory and estrogenic
activity (Sharifi-Rad et al. 2021).
This study was designated to demonstrate the antioxidant effect as
well as therapeutic impact of licorice roots extract on pituitary adrenal axis
hormones in AI induced by glucocorticoids administration in female rats.
Materials and methods
Ethical Approval
This study was performed in the animal house at National Research
Centre (NRC), under the ethical approval number 13070105-1 obtained by NRC
Ethics Committee.
Animals
For this experiment, forty male rats (2-3 months) aged weighing
180-210 grams. The animal was housed in ventilated cages (10 rats /cages)
beneath optimum circumstances in the animal house comprising unlimited water
and free use of a commercial diet. The animals were let ten days to acclimatize
to the lab environment.
Experimental design
The Forty male rats were sectioned randomly into four groups. The
first was negative control group (G1); normal saline was administered orally to
Rats. The second was positive control group (G2); a dose of 50 mg/kg of
Hydrocortisone Sodium was administered intra-peritoneal to rats for 7 days. The
third therapeutic group (G3); the same of group 2 and then followed by oral
administration of licorice roots extract by100 mg /kg for14 days. The last
fourth group (G4); rats were orally given 100 mg/kg of licorice roots extract
for 21 days. Blood samples were taken from cardiac punctures at the end of the
experiment and placed into gel tubes for the analyses.
Hormonal and biochemical assay
The serum ACTH, cortisol, CRH hormones,
11beta-HSD enzyme, and malondialdehyde (MDA) were measurement by using
commercial kits (RayBiotech / USA). Serum sodium and potassium levels
were measured by colorimetric method according to (Frezzotti et al., 1996).
Statistical analysis
The data were statistically analyzed
using the ANOVA in the computerized SPSS program version 24.0.
Results
Influence of licorice roots extracts
on ACTH, Cortisol, and CRH hormones in adrenal incapability female Rats.
The
current study demonstrated a significant reduction (P<0.05) in serum
concentration of ACTH, cortisol, and CRH hormones in the G2 group administered
hydrocortisone paralleled to the control G1 (Table 1). Licorice roots extract treatment
induced a significant elevation (P<0.05) in G3 (therapeutic) group compared
with the G2 group. While, the ACTH and the cortisol values in G3 group have
significant variations (P<0.05) compared to the control G1. On the other
hand, no significant differences between G4 and the control group in all three
hormones, as shown in (Figure. 1).
Table 1: influence of licorice roots
extract on serum ACTH, cortisol, and CRH concentration in adrenal incapability
in male rats.
|
Group |
Treatment |
ACTH (Pg/ml) |
Cortisol (nmol/L) |
CRH (Pg/ml) |
|
(G1) |
Control Negative |
93.76 ±7.34 |
62.52 ±4.18 |
7.42 ±0.25 |
|
(G2) |
Control Positive |
46.57 ±2.64 |
30.11 ±4.88 |
4.82 ±0.4 |
|
(G3) |
Therapeutic |
70.21 ±3.05 |
41.35 ±9.50 |
6.2 ±1.25 |
|
(G4) |
Licorice
roots extract |
100.23 ±4.56 |
67.41 ±7.9 |
6.67 ±0.55 |
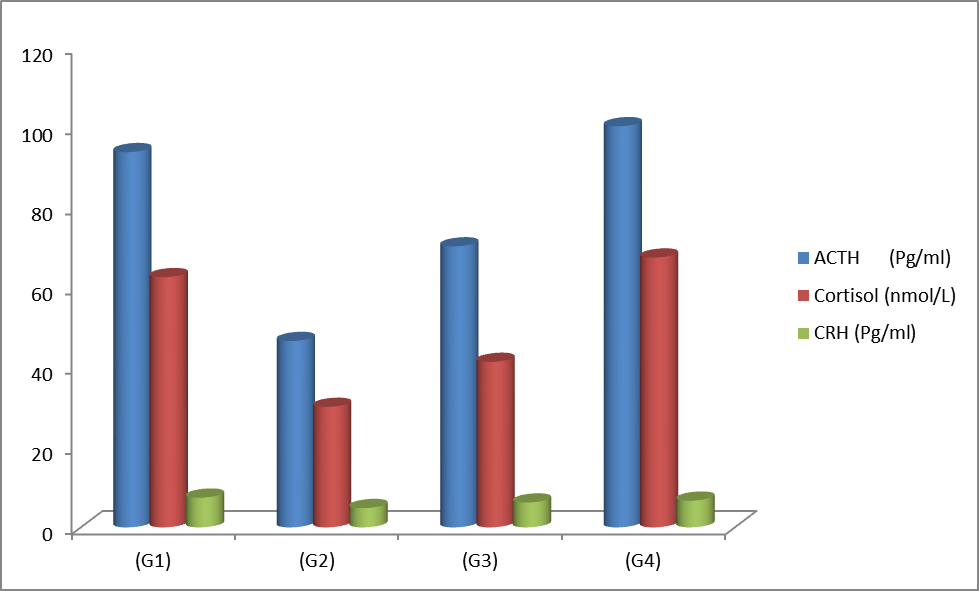
Figure 1: Impact of licorice roots extract on serum ACTH, cortisol,
and CRH concentration in adrenal incapability in male rats. G1: received normal
saline, G2: received hydrocortisone, G3: received hydrocortisone then licorice
roots extract, G4: licorice roots extract only.
Influence of licorice roots extracts on 11beta-HSD enzyme and MDA
in adrenal incapability in male rats
The obtained data displayed that the
mean value of 11 β -HSD enzyme has diminished significantly (p˂0.05) in G2
compared with the control. The effect of daily administration of licorice roots
extract indicates a significant decrease (p˂0.05) in the G3 and G4 groups
compared with the control group G1 as shown in (Table 2) and (Figure 2).
Table 2: Influence of licorice roots
extract on serum 11beta-HSD enzyme and MDA concentration in adrenal
incapability in male rats.
|
Group |
Treatment |
11beta-HSD ng/dl |
MDA nmol/dl |
|
(G1) |
Control
Negative |
0.413
±0.05 |
0.179 ±0 .03 |
|
(G2) |
Control
Positive |
0.135
±0.03 |
3.54±0.688 |
|
(G3) |
Therapeutic |
0.140
±0.15 |
0.65 ± 0.18 |
|
(G4) |
Licorice
roots extract |
0.110
±0.26 |
0.182 ±0.03 |
On the other hand, the results in Table (2) revealed that a significant
elevation (P≤0.05) in serum MDA in the adrenal incapability (G2) compared to
the control G1, G3 and G4 groups. Hence, there were no significant variations
G3 and G4 groups after being treated with licorice roots extract compared to
the control G1(Figure 2).
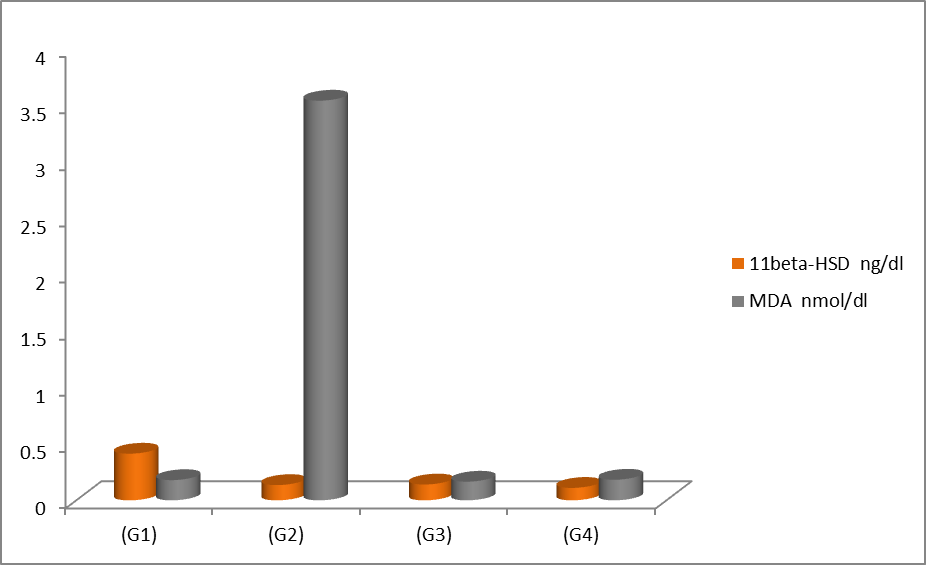
Figure 2: Impact of licorice
roots extract on 11-β HSD enzyme and MDA in adrenal incapability in male rats.
G1: received normal saline, G2: received hydrocortisone, G3: received
hydrocortisone then licorice roots extract, G4: licorice roots extract only.
Influence of licorice roots extracts
on serum sodium and potassium
concentration in adrenal incapability in male rats
Table 3: Influence of licorice roots extract on serum sodium and
potassium concentrations in adrenal incapability in male rats.
|
Group |
Treatment |
Serum sodium mg/dl |
Serum
potassium mg/dl |
|
(G1) |
Control Negative |
142.23 ±0.56 |
5.23 ±0.56 |
|
(G2) |
Control Positive |
130.23 ±1.33 |
6.47 ±0.44 |
|
(G3) |
Therapeutic |
145.27 ±1.24 |
5.42 ±0.33 |
|
(G4) |
Licorice roots extract |
170.25 ±2.16 |
5.33 ±4.56 |
Our outputs in table (3) exhibited
that there was a significant elevation in serum sodium accompanied with
significant reduction in potassium levels in groups administered with GA
compared to control group. Contrarily, the results showed a significant decreasing
in serum sodium accompanied with rise in potassium levels in G2 which
constituted adrenal incapability compared to control group (G1) as shown in
figure (3).
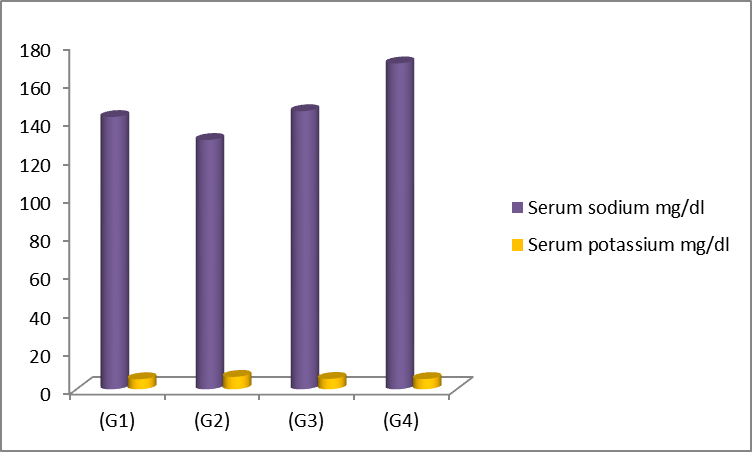
Figure 3: Impact of licorice roots extract on serum
sodium and potassium concentration in adrenal incapability in male rats. G1:
received normal saline, G2: received hydrocortisone, G3: received
hydrocortisone then licorice roots extract, G4: licorice roots extract only.
Discussion
Animal models are often employed to
understand the pathophysiology of glucocorticoids caused adrenal incapability
and to test pharmacological remedy. In our study hydrocortisone
intra-peritoneal administration for 7 days induces a significant reduction in
the serum concentration of ACTH, cortisol and CRH. A parallel finding from Téblick et al., (2022) elucidated that exogenous
hydrocortisone causes a negative effect on the hypothalamus pituitary gland
axis, leading to a decreasing of CRH and ACTH, accompanied with the reduction
in cortisol production. The pathophysiology of glucocorticoids is multi-reason,
and it probably act via suppressed CRH, dopaminergic and central noradrenergic
system, due to chronic suppression of HPA axis, and rise in cytokines, and
prostaglandins (Kao et al., 2014) and (Improda et
al., 2024).
The administration of licorice roots
extract to groups treated with hydro- cortisone result in a significant
elevation in ACTH, cortisol, and CRH levels and overcome the low levels that
occurred in the adrenal incapability group (G2). Our data uphold the earlier
studies (Lin et al., 2012) which administered glycyrrhetinic
acid in the male rats which exposed a higher concentration of cortisol levels.
The authors assumed this adrenal incapability condition attributed to the
suppressive effect on 11β-HSD.
On the other side, our results
revealed that the licorice roots extract treated groups were a rise in the
11β-HSD concentration than the group administered with hydrocortisone only.
This enzyme is important in the conversion process of cortisol to other derivatives
and is substantial for regulating the glucocorticoid and mineralocorticoid
receptors. The typical suppression of 11-HSD by, bioactive constituents of
licorice is endogenous steroidal substances acting as glycyrrhetinic
acid-like agents that block 11-HSD and enable glucocorticoid-induced
mineral-receptors and glucocorticoid-receptors stimulation, may employ as
competitive substrates, while others only act as suppressors (Bailly, and Vergoten 2020, Matchanov et al.,
2022).
Moreover, glycyrrhetinic
acid prevents the conversion of cortisol to inactive cortisone by inhibiting
11β-HSD (Hardy et al., 2013). Certain investigations displayed the selective
suppression of glycyrrhizic acid as it was mentioned
that 18α- glycyrrhizic acid preferentially
selectively inhibits type 1- 11β-HSD, while 18β-GA preferentially suppress type
2-11β-HSD (Sakoda et al., 2024).
Lipid peroxidation is an important
concern induced by free radicals within an organism. Malondialdehyde is
considered a by-product of the polyunsaturated fatty acids peroxidation in the
cells. An elevation in free radicals induces excessive output of MDA. Commonly,
malondialdehyde level is employed as an indicator for oxidative stress and the
existence of antioxidants (Tyagi, et al., 2015, Alobaidi,
2024). Extracted from our data, by glucocorticoid injection in G2, adrenal
oxidative stress was asserted by significant elevation estimating of MDA. The
glucocorticoids enhance the output of free radicals as relative oxygen species
(ROS), which is a reason of adrenal injury by oxidizing cell membrane lipids,
DNA damage and protein denaturation (Flaherty et al., 2017). Whereas, treatment
with licorice roots extract mitigated the glucocorticoid-caused oxidative
damage by decreasing MDA levels. This could be explicated by the efficiency of
licorice roots extract to amend certain enzymes embraced in inflammation, oxidative
stress, and the inhibition of some pro-inflammatory interleukins, safeguarding
cells from destruction induced by inflammation or ROS (Ageeva
et al., 2022). Our results coincided with (Feng et al., 2013; Galanis et al., 2019; Wang et al., 2022) who reported that
licorice minimized serum levels of malondialdehyde (MDA) in rats. Additionally,
many researches correlate the anti- oxidant merit of licorice with its
anti-inflammatory ability. It has been illustrated that licorice inhibits the
formation of nitric oxide and inflammatory interleukins together with other
components of licorice extract (Li et al., 2011; Richard, 2021).
Our result revealed a marked
elevation in serum sodium associated with significant decreasing in potassium
levels in groups administered with licorice roots extract paralleled to control
group and can restored the significant decreasing in serum sodium and rise in
potassium levels in G2 which constituted adrenal incapability.
Hejazi et al. (2017) developed in
vitro and in silico evaluation based pharmacokinetic (PBPK) model in rat to
assess administration of licorice roots extract and reported increase in sodium
and reduction in potassium levels. On other sight, It was demonstrated that
oral taken 100 mg/kg per day licorice roots extract did not influence serum
electrolyte as potassium and sodium levels, referring no onset of undesirable
edema (Fernando et al. 2014).
Conclusion
Extracting from our findings, we can
conclude that licorice roots extract components have a beneficial impact in the
treatment and protection of hormonal, biochemical and oxidation alterations in adre- nal insufficiency rats
induced by glucocorticoid.
References
Ageeva, A.A., Kruppa, A.I., Magin,
I.M., Babenko, S.V., Leshina,
T.V., and Polyakov, N.E. New aspects of the antioxidant activity of
glycyrrhizin revealed by the cidnp
technique. Antioxidants, 11(2022):1591.
Bailly,
C., and Vergoten, G. Glycyrrhizin: An alternative
drug for the treatment of COVID-19 infection and the associated respiratory
syndrome? Pharmacology&therapeutics, 214(2020):107618.
Borresen, S.W., Klose, M., Glintborg, D., Watt,
T., Andersen, M.S., and Feldt-Rasmussen, U. Approach to the patient with
glucocorticoid-induced adrenal incapibility. The
Journal of Clinical Endocrinology and Metabolism, 107(2022): 2065–2076.
Díaz-Castro,
F., Monsalves-Álvarez, M., Rojo, L.E., Del Campo, A.,
and Troncoso, R. Mifepristone for Treatment of
Metabolic Syndrome: Beyond Cushing's Syndrome. Frontiers in
pharmacology, 11(2020):429.
Feng,
L., Zhu, M.M., Zhang, M.H., Wang, R.S., Tan, X.B., Song, J., Ding, S. M., Jia,
X.B., and Hu, S.Y. Protection of glycyrrhizic acid
against AGEs-induced endothelial dysfunction through inhibiting RAGE/NF-κB pathway activation in human umbilical vein endothelial
cells. Journal of Ethnopharmacology, 148(2013): 27–36.
Fernando,
H.A., Chin, H.F., Ton, S.H., Abdul Kadir, K. Stress and its effects on glucose
metabolism and 11β-HSD activities in rats fed on a combination of high-fat and
high-sucrose diet with glycyrrhizic acid. J Diabetes
Res. 2013 (2013):190395.
Flaherty,
R. L., Owen, M., Fagan-Murphy, A., Intabli, H.,
Healy, D., Patel, A., Allen, M.C., Patel, B.A., and Flint, M.S. Glucocorticoids
induce production of reactive oxygen species/reactive nitrogen species and DNA
damage through an iNOS mediated pathway in breast
cancer. Breast Cancer,Research
BCR, 19(2017):35.
Fredrick,
F.C., Meda, A.K.R., Singh, B., and Jain, R. Critical illness-related
corticosteroid incapability: latest pathophysiology and management
guidelines. Acute and Critical Care, 39(2024):331–340.
Frezzotti, A., Margarucci, A.M., Coppa, G., De
Sio, G. An evaluation of the Ektachem serum lithium
method and comparison with flame emission spectrometry. Scand
J Clin Lab Invest. 56(1996):591-6.
Galanis, D., Soultanis, K., Lelovas,
P., Zervas, A., Papadopoulos, P., Galanos, A., Argyropoulou, K., Makropoulou,
M., Patsaki, A., Passali,
C., Tsingotjidou, A., Kourkoulis,
S., Mitakou, S., and Dontas,
I. Protective effect of Glycyrrhiza glabra roots extract on bone mineral
density of ovariectomized rats. BioMedicine, 9(2019):8.
Hardy,
R.S., Seibel, M.J., and Cooper, M.S. Targeting 11β-hydroxysteroid
dehydrogenases: a novel approach to manipulating local glucocorticoid levels
with implications for rheumatic disease. Current Opinion in
Pharmacology, 13 (2013): 440–444.
Hejazi,
I.I., Khanam, R., Mehdi, S.H., Bhat, A.R., Moshahid
Alam Rizvi, M., Islam, A., Thakur, S.C,, Athar, F. New insights into the
antioxidant and apoptotic potential of Glycyrrhiza glabra L. during
hydrogen peroxide mediated oxidative stress: An in vitro and in silico
evaluation. Biomed Pharmacother. 94 (2017):265-279.
Improda, N., Chioma, L., Capalbo,
D., Bizzarri, C., and Salerno, M. Glucocorticoid treatment and adrenal
suppression in children: current view and open issues. Journal of
endocrinological investigation, (2024).
10.1007/s40618-024-02461-9. Advance online publication.
Kao,
T.C., Wu, C.H., and Yen, G.C. Bioactivity and potential health benefits of
licorice. Journal of Agricultural and Food
Chemistry, 62(2014):542–553.
Lewis,
A., Thant, A. A., Aslam, A., Aung, P.P.M., and Azmi, S. Diagnosis and
management of adrenal incapability. Clinical medicine (London,
England), 23(2023):115–118.
Li,
Y.J., Chen, J., Li, Y., Li, Q., Zheng, Y.F., Fu, Y., and Li, P. Screening and
characterization of natural antioxidants in four Glycyrrhiza species by liquid
chromatography coupled with electrospray ionization quadrupole time-of-flight
tandem mass spectrometry. Journal of
Chromatography. 1218(2011):8181–8191.
Matchanov, A.D., Esanov, R.S., Renkawitz,
T., Soliev, A.B., Kunisch,
E., Gonzalo de Juan, I., Westhauser, F., and Tulyaganov, D.U.. Synthesis, Structure-Property Evaluation
and Biological Assessment of Supramolecular Assemblies of Bioactive Glass with Glycyrrhizic Acid and Its Monoammonium Salt. Materials
(Basel, Switzerland), 15(2022):4197.
Nachawi, N., Li, D., and Lansang, M.C.
Glucocorticoid-induced adrenal incapibility and
glucocorticoid withdrawal syndrome: Two sides of the same coin. Cleveland
Clinic Journal of Medicine, 91(2024):245–255.
Richard,
S.A. Exploring the Pivotal Immunomodulatory and Anti-Inflammatory Potentials of
Glycyrrhizic and Glycyrrhetinic
Acids. Mediators of Inflammation, 2021 (2021): 6699560.
Sakoda,
R., Ishiuchi, K., Yoshino, T., Tsunoo,
Y., Namiki, T., Ogawa-Ochiai,
K., Minamizawa, K., Fukunaga, K., Watanabe, K., and
Makino, T. 3-epi-18β-glycyrrhetinic
acid or its glucuronide, the metabolites of glycyrrhizinic
acid with individual differences, correlated with diagnostic marker for
licorice-induced pseudoaldosteronism in
humans. Drug metabolism and disposition: the biological fate of
chemicals, 52(2024):1407–1416.
Sharifi-Rad,
J., Quispe, C., Herrera-Bravo, J., Belén, L.H., Kaur, R., Kregiel,
D., Uprety, Y., Beyatli,
A., Yeskaliyeva, B., Kırkın,
C., Özçelik, B., Sen, S., Acharya, K., Sharopov, F., Cruz-Martins, N., Kumar, M., Razis, A.F.A., Sunusi, U., Kamal,
R.M., Shaheen, S., Suleria, H.A.R. Glycyrrhiza Genus:
Enlightening Phytochemical Components for Pharmacological and Health-Promoting
Abilities. Oxidative Medicine and Cellular
Longevity, 2021(2021): 7571132.
Téblick, A., Gunst, J., and Van den Berghe, G. Critical Illness-induced Corticosteroid
Insufficiency: What It Is Not and What It Could Be. The Journal of
Clinical Endocrinology and Metabolism, 107(2022):2057–2064.
Wang,
H., Zhang, B., Dong, W., Li, Y., Zhao, L., and Zhang, Y. Effect of Diammonium
glycyrrhizinate in improving focal cerebral ischemia-reperfusion injury in
rats through multiple mechanisms. Dose-response: a publication of
International Hormesis Society, 20(2022): 1559325.
Yaw,
H.P., Ton, S.H., Chin, H.F., Karim, M.K., Fernando, H.A., and Kadir, K.A.
Modulation of lipid metabolism in glycyrrhizic
acid-treated rats fed on a high-calorie diet and exposed to short or long-term
stress. International Journal of Physiology, Pathophysiology and
Pharmacology, 7(2015): 61–75.