Microbial Infections of Pet
Animals Urinary Tract: Review Article
*Abouelhag H. A.
Microbiology and Immunology Dept., National Research Centre, Dokki,
Giza, Egypt, 12622
*Corresponding author: Abouelhag H. A. Email:
deabouelhag@yahoo.com
Received: 25-04-2025, Accepted: 05-05-2025, Published online:
23-05-2025
DOI: https://doi.org/10.33687/ricosbiol.03.05.34
Abstract
Microbial urinary tract infections
(UTIs) are reported to be between the second and third most common reasons for
antimicrobial use in pet animals, dogs and cats, representing 12% of all
antibiotic prescriptions. UTI refers to microbial colonization of any portion
of the urinary system that is normally sterile. The distal urethra is not
sterile; it has a normal flora. UTIs are often caused by bacterial organisms
that are part of the microflora of the intestinal tract. UTI is usually caused
by one single bacterial species. Predominant bacterial species were Gram-ve
bacteria, as Escherichia coli constituted the major isolated species
(about 40-50%), followed by Proteus spp., Pseudomonas aureogenosa,
Klebsiella spp., and Enterobacter spp. Gram-positive bacteria such
as Staphylococcus spp., Enterococcus spp., and Streptococcus
spp., Leptospira and Mycoplasma spp. Additionally, fungal and
viral causes play a reside role to some extent in UTIs. Diagnosis of UTIs in
pets is based on clinical signs, urine analysis, and bacterial culture results
obtained from urine samples collected, preferably using cystocentesis. The
empiric antimicrobial treatments are often administered in the presence of
clinical signs as they affect a broad spectrum of bacterial etiology associated
with UTIs. Antimicrobial therapy is indicated in most cases while awaiting
culture and susceptibility results to overcome the condition.
Multidrug-resistant bacteria are an alarming development with significant
public and pet health ramifications. Natural alternative methods can be useful
as supplemental therapy choices and are much required. Cranberry is frequently
used to prevent UTIs in older male dogs, but more research is needed.
Prophylactic antibiotic medication, particularly for non-neutered male dogs,
has not yet been shown to be significant; however, it may be of help in some
cases. This work aimed to supply the researchers and veterinarians with a wider
point of view about urinary tract infections in pet animals.
Keywords: Pet animals, Urinary tract
infections, UTIs, Companian animals, Diagnosis, Treatment and follow up.
INTRODUCTION
Urinary tract infections (UTIs)
are a common concern in pet animal practice as sequelae of compromised host
defense mechanisms and a virulent microbe adhere, multiply, and persist in a
portion of the urinary tract. The prevalence of UTIs in the dog over its
lifetime has been reported to be 14%, and in cats, it has been reported to be
between 3% to 19% (Pereira et al., 2024).
There are numerous factors
thought to impact the risk of UTI in species, comprising sex, age,
comorbidities, and functional abnormalities of the urinary tract. Host defenses
include normal micturition, anatomic structures, the mucosal barrier,
properties of urine, and systemic immunocompetence. (Amphaiphan et al.,
2021).
UTIs may involve more than one
anatomic location, and the infection should be categorized as upper urinary
tract (kidneys and ureters) versus lower urinary tract (bladder and urethra).
Most bacterial UTIs occur as a consequence of ascending migration of pathogens
through the genital tract and urethra to the bladder, ureters, and one or both kidneys
(Tigabie et al., 2025).
Frequently, UTIs are induced by
bacteria, fungi and viruses. Most of the UTIs in dogs and cats (~75%) involve a
sole agent, 20% two co-infecting species, and approximately 5% are caused by 3
mixed species, with Escherichia coli (E. coli)being responsible
for up to half of the infections in dogs. This Gram -ve organisms are also the
most common pathogen in cats (60%), with Staphylococcus felis (S.
felis)being the most common Gram +ve in that species, followed by 20% of
other Gram-positive cocci, in addition to Leptospira spp. (Deprey et al., 2021) and Mycoplasma
spp. (Alves et al., 2023). Fungal UTIs is uncommon and occurs usually
because of temporary or permanent breaches in immunity of the lower UTIs. Candida
albicans is the most commonly followed by Candida glabrata and Candida
tropicalis. Also, other fungi may include as Aspergillus spp., Blastomysis
spp., and Cryptococcus spp. (Reagan et al., 2019; Sender et al.,
2024). Viral-induced diseases in pets are increasingly determined, particularly
of the upper urinary tract, as canine adenovirus type I, herpesvirus, as well
as feline coronavirus and leukemia virus (Kruger et al., 2011). A better
understanding of the defense mechanisms of the urinary tract, the behavior of
uropathogenic bacteria, and a rising awareness of the dangers of antimicrobial
resistance have led to alterations in the recommendations for diagnosis and treatment
of UTI in dogs and cats (Grant et al., 2021). So the emergence of
multidrug-resistant bacteria (MDR) isolates resistant to three or more
antimicrobial categories’ implemented in UTI in dogs and cats creates questions
about the role of companion animals as potential reservoirs of resistant
bacteria and has been reported as a serious public health problem (Smoglica et
al., 2022).
Given these challenges, there is
a critical need to explore alternative treatments that can effectively combat
MDR urinary pathogens. These types of treatments are receiving increasing
attention in the treatment of UTIs, especially uncomplicated clinical
conditions (Biasibetti et al., 2019).
The current article aimed to
discusse the microbial urinary tract infections in pet animals; their types,
the risk factors for complicated conditions, the major associated species, the
standards for diagnosis, and different antimicrobial and non-antimicrobial
remedy approaches. The zoonotic risks and public health implications associated
with MDR UTIs in dogs and cats are addressed.
TYPES OF UTI IN DOGS
AND CATS
UTIs in
pets can be either: i) Simple or uncomplicated (sporadic cystitis): No
predisposing factors or other diseases present; ii) Complicated or recurrent:
Seen in pets with underlying medical conditions or predisposing causes; pets
with more than three UTIs in the past 12 months (Smee et al., 2013). The
2019, International Society for Companion Animal Infectious Diseases revised
the classifications of UTI. The revised classification has 3 diagnoses:
subclinical bacteriuria, sporadic cystitis, and recurrent UTI (RUTI) (Weese et
al., 2019).
Predisposing
or risk factors for different types of UTIs were correlated very often to
several urinary disorders; with the occurrence and the development of urinary
bladder stones, insufficient protection of the urogenital tract against
external influences due to immunosuppressive therapy (e.g. hyperadrenocorticism
and diabetes mellitus) are important risk factors contributing to UTIs in pets.
From the anatomical point of view, it is necessary to mention that the urethra
in the females of dogs and cats is shorter and wider in comparison to males and
this is the reason why urethritis develops more often in females than in males.
UTIs are more common in older female dogs (>7 years) (Kocúreková et al.,
2021).
The significant
association of breeds with the presence of UTIs was reported, where Golden
Retrievers were found to be more likely to be positive for E. coli/K.
pneumoniae than other pure breeds or mixed breeds, according to a
statistical analysis. (Facchin et al., 2025).
2.
BACTERIAL CAUSES OF
UTIS IN DOGS AND CATS
Bacterial urinary tract
infections are one of the more common infections in dogs. Approximately 14% of
the canine population afflicted by a UTIs. Although they often affect older
female canines (>7 years) (Hernando et al., 2021).
2.1.
Escherichia coli
E. coli is the most frequently isolated
pathogen, with a prevalence ranging from 35% to 64%, fundamentally, most UTIs are caused by what is termed
“extraintestinal pathogenic E. coli”
(ExPEC) which are belong to phylogenetic group B2 (generally containing more
virulence factors or genes) and to a lesser extent group D. These groups of
organisms are phylogenetically distinct from commensal and intestinal E.
coli, which predominantly belong to groups A and B1 (Govindarajan
et al., 2024).
Specific uropathogenic characteristics and virulence factors
(VFs) are required for bacterial strains to initiate an infection, regardless
of the presence of a competitive colonization advantage in the gut. VFs of
importance to uropathogenic E. coli (UPEC) include capsular factors,
cytotoxins, invasion factors, siderophores and related transport systems, as
well as adhesins that mediate binding to the renal tubule (P, S and F1C
fimbriae) and bladder urothelium (Type I fimbriae). This is likely to be of
particular importance in dogs with intact urinary tract defense mechanisms (Fig. 1). However, phylogenetic group A and B1 E.
coli, whilst generally possessing fewer VFs than group B2 and D E. coli,
may also initiate UTI in dogs if host defenses are compromised (Halaji
et al., 2022).
E. coli that
colonize the urinary tract can protect themselves from the harsh bladder
environment by forming biofilms. These biofilms promote persistence leads to
chronic and recurrent UTIs (Ballash et al., 2022). E. coli are
assumed a global threat because of the lowering options for antimicrobial
therapy. Pets could be a reservoir of multidrug resistanate (MDR) E. coli,
and the households owning pets had increased (Teng et al., 2023).
In a
recent Italian study, urine samples collected from 133 dogs manifested at least
one of UTI clinical signs and admitted to the Veterinary Teaching Hospital of
Milan. Out of these, 28 E. coli strains were found, with 60. 7%
producing biofilms, 25% being multidrug-resistant, and 3. 6% harboring ESBL
(Facchin et al., 2025). An investigation study across 12 European
countries supported E. coli as the most frequently isolated bacterium in
dogs; 46.9% and 61.2% in cats (Temmerman et al., 2024). The incidence
of E. coli was 45.58% in study included 2,583 urine samples from
dogs suspected of UTIs with high prevalence of resistance to ampicillin
(31.42%) (Yudhanto et al., 2022). Hernando et al., (2021)
determined E. coli in UTI dogs as (1333/2942; 45.3%) in Spanish survey.
While, Punia et al., (2018) reported less incidence of E. coli;
(29.62%) in urine samples collected from 35 dogs suspected of UTI, in India. In
Egypt, Hakim et al., (2024a) determined 34 and 8 E. coli isolates
out of 81canine (41.97%) and 38 feline (21.05%) urine samples with notable
resistance against imipenem and loading of blaNDM-1gene was responsible
for resistance against carbapenems. Farag et al., (2024) mentioned that E.
coli was the most common pathogen isolated from 146 dogs (46.4 %) and 162
cats (66.7 %) suffered from lower urinary tract disorders.
2.2.
Leptospira spp.
Leptospirosis
is caused by pathogenic spirochetes of the genus Leptospira, which
colonize the renal tubules where they reproduce before being excreted via
urine. Contaminated water with infected urine is the source of leptospirosis
infection, where Leptospira can enter the bodies of mammalian hosts via
lacerations in the skin, contact with mucosa, conjunctiva, and inhalation of
aerosols as shown in figure (1). Dogs may have an asymptomatic form or may
suffer from a wide range of clinical manifestations, including hepatic, renal
failure and severe pulmonary hemorrhage (Schuller et al., 2015).
Formerly, it was thought that domestic cats were resistant to leptospirosis
infection. However, published reports on feline leptospirosis concluded that
cats are exposed to Leptospira and may play a role in the epidemiology of the
disease (Palerme et al., 2019). A study investigated Leptospira
spp. prevalence in 112 cats from southern Italy, the data revealed detection of
6 serovars in 15.3% (17/112) of tested cats so can represent an additional
reservoir or sentinel for a risk of infection (Donato et al., 2022).
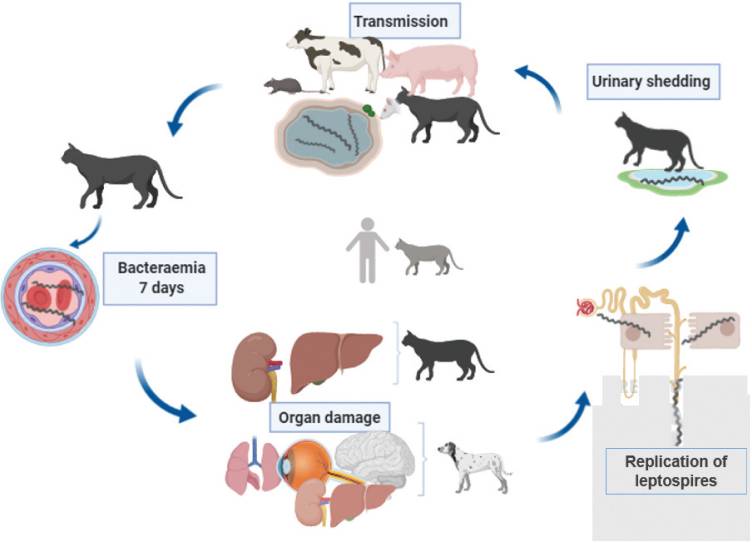
Figure (1) Proposed pathogenesis of leptospirosis in
cats and dogs. Replication of leptospires occurs in the kidney leading to
urinary bacterial shedding (Murillo et al., 2020).
A
cross-sectional study in Reunion Island determined cats as a part of the
maintenance community of different strains of Leptospira spp. The
prevalence of Leptospira infection in 92 samples of stray and domestic
cats has been studied using serological and molecular detection. The results
revealed a seroprevalence of 37.0% (34/92) (cut-off 1:40), while using PCR;
28.6% (12/42) of stray cats were tested positive and Leptospiral DNA was
detected in renal tissue, urine and blood. The study confirmed that renal carriage
and urinary shedding are possible, especially in stray cats which can be
considered potential actors within the maintenance community of Leptospira
in Reunion Island (Holzapfel et al., 2021). Also, a longitudinal study
was performed using a multidisciplinary approach for the identification of
chronically infected stray and sheltered dog populations in São Paulo, Brazil.
A total of 123 dogs from three populations were included. Asymptomatic L.
santarosai infection was observed in all populations studied, suggesting a
possible role of dogs in the chain of transmission of this leptospiral species
(Miotto et al., 2018). Another study of pet leptospirosis detection in
Algeria, was conducted in the urines of stray dogs and cats. The results
revealed that 5/104 (4.8%) canine urine samples (asymptomatic mixed-breed dogs)
were positive while all of the 107 cat urine samples were negative. The
confirmed L. interrogans prevalence was significantly higher in dogs
aged < one year (16.46% - 29.41%) than in adults (Zaidi et al.,
2018). On the other hand, Delaude et al. (2017) stated that in spite of
human leptospirosis remains rare in Switzerland, the incidence of canine
leptospirosis is unusually high compared to other European countries.
Leptospirosis
pulmonary hemorrhage syndrome (LPHS) in dogs and cats has poor prognosis due to
acute respiratory failure and dyspnea, leading to death. Cats with mild
clinical signs respond well to antimicrobial therapy, while those with chronic
leptospirosis developed permanent renal damage (Murillo
et al., 2020).
2.3. Mycoplasma spp.
Mycoplasmas
are considered to be part of the normal microbial flora of canine mucosal
membranes. However, some of these mycoplasmas may causes UTI by ascending
migration from the lower urinary or genital tract. Hemmatzadeh (2019) reported the first report of detection of Ureaplasma
canigenitalium in an English Cocker Spaniel dog with the history of UTIs
and chronic renal insufficiency in Australia. Mycoplasma canis was
isolated from four of 100 (4%) urine samples obtained by cystocentesis from 100
dogs with symptoms of lower urinary tract disease. Also, Mycoplasmas identified
as M. canis were isolated from nine dogs with clinical signs of
urogenital disease in Norway over 20 months (L'Abée-Lund et al., 2003).
2.4.
Other Bacterial
Species
In
Italy, urine samples collected from 133 dogs at Veterinary Teaching Hospital of
Milan showed positive microbiological culture of K. pneumoniae
isolates, (4.51%) (Facchin et al.,
2025).
European
survey determined the causative bacteria of canine UTIs next to E. coli;
Staphylococcus intermedius and Proteus mirabilis (13.1%). The
frequent Gram +ve isolates were Streptococcus spp. (8.3%),
Enterococcus spp. (8.0%) and S. aureus was (0.9%).
Gram-negative bacteria included Klebsiella spp. (4.0%), Pseudomonas
aeruginosa (3.6%) and finally Pasteurella spp. In second period
(0.5%). On the other hand, among examined feline urine samples,
coagulase-negative Staphylococci were the second-most frequently
isolated species (13.2%), including the specific pathogen Staphylococcus
felis. Enterococcus spp. was (17.7%) and S. aureus (4.2%).
The other Gram-negative pathogens; Proteus spp., Pseudomonas
spp., Klebsiella spp., and Pasteurella spp. were 5.3%, 4.2%, 1.6%
and 1.6%, respectively (Temmerman et al., 2024).
A study
aimed to bring new insights into the current bacterial urinary tract infections
in companion animals scenario of Portugal showed 5306/17472 (30.4%) +ve
bacterial culture. Of the culture-positive samples, 5224 (96.6%) were pure
cultures and 82 (3.2%) had mixed growth. Other E. coli bacteria were Proteus
mirabilis (11%), Enterococcus faecium (5.2%) and
Staphylococcus pseudintermedius (4.3%), as shown in Figure (2) (Garcês, et
al., 2022).
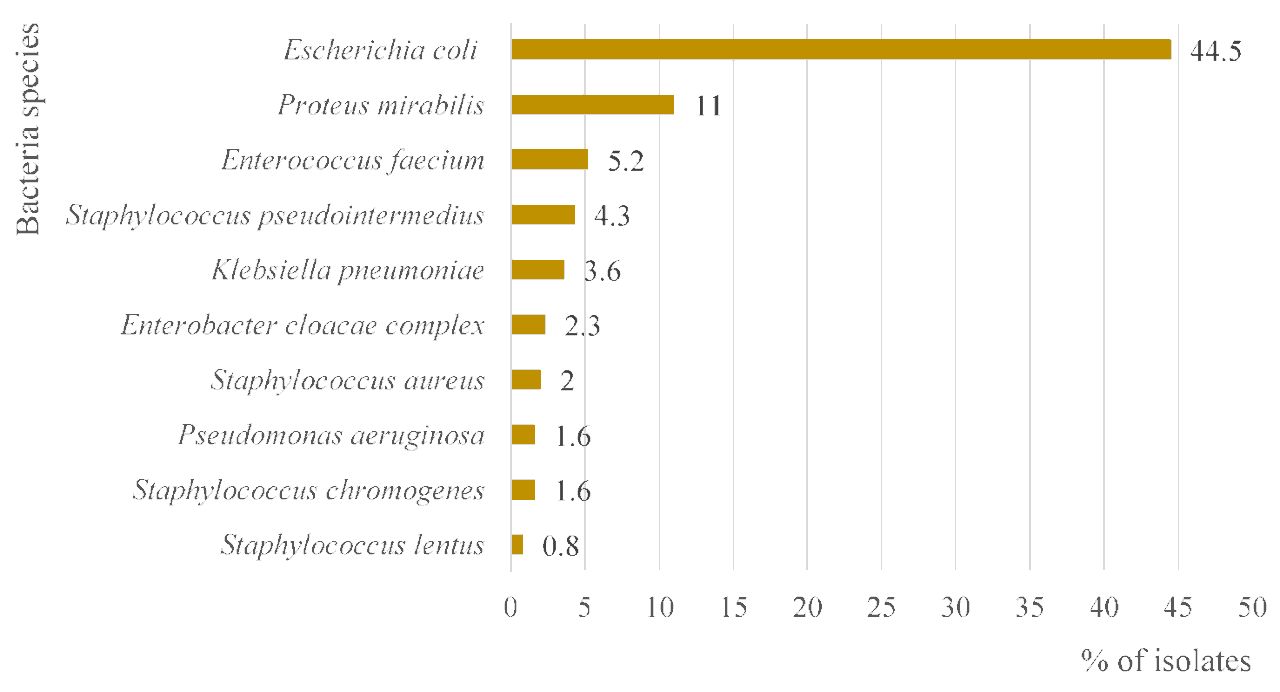
Figure 2: Bacteria species that predominate in the 5306
isolates with positive culture grown from urine samples from dogs and cats
submitted to the INNO veterinary laboratory between 2017 and 2021 (Garcês et al., 2022).
In
Egypt, hypervirulent type K. pneumoniae (hvKp) isolates were recovered
from canine urinary samples in Al Qalyubia and Giza Governorates by a rate of
2% (Soliman et al., 2024). While. Farag et al., (2024) recorded
that the second prevalent species isolated were Proteus spp. in canine
isolates (16.1 %) and Klebsiella spp. in feline isolates (14.3 %). Staphylococcus
spp. was isolated from canine cases only with the detection of
methicillin-resistant Staphylococcus pseudintermedius (MRSP) strains at
3.6 %.
3. MYCOTIC
URINARY TRACT INFECTIONS
Mycotic
infections that frequently affect the urinary tract of dogs and cats are
generally flourished when animal’s immunological state become lower. Most of
fungal UTIs are caused by an overgrowth of Candida spp. Although candida
yeasts are normally present in the body as well as on the skin circumstances,
it can disrupt the natural functioning of the body, when this occurs in the
lower urinary tract it causes an infection which can become progressively. Fungal
infections of the lower urinary tract often are asymptomatic, and may be
uncovered when diagnosing another issue or during regular veterinary check-ups.
Opportunistic mycoses in dogs and cats can result in a wide variety of
symptoms, from localized infections to catastrophic systemic illnesses. Such
fungi like Microsporum canis and Sporothrix brasiliensis may be
important zoonotic agents (Eissa, 2023).
3.1.
Candida species
Candiduria
is the most commonly reported manifestation of candidiasis in the veterinary
literature as shown in figure (3). In a retrospective study of urinary tract
infections in dogs; risk factors for candiduria in pet animals were reported;
comorbidities involved diabetes mellitus, antibiotic use in the preceding
30 days, immunosuppression, and lower urinary tract illness (Sykes et al.,
2014).
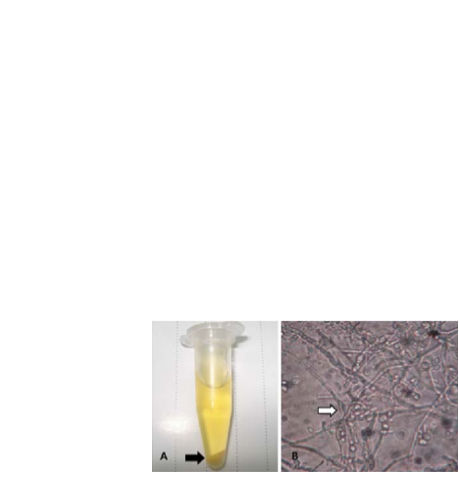
Figure 3:
Urine sample (A) Cloudy sediment (black arrow) after centrifugation of urine.
(B) Microscopic examination found pseudohyphae and budding yeast (white arrow)
indicating Candida infection. 1,000×g (Sung et al., 2017).
Eighteen
dogs belonged to 4 mixed breeds with candiduria without suspected systemic
infection were identified. Three species of Candida were isolated, C.
albicans, and C. tropicalis, and then C. glabrata.
Ages ranged from 1‐14 years. of age (median 7 years.). Antibacterial drug
administration within the 30 days before diagnosis was recorded in 15 (83%)
dogs. Potential causes of immunosuppression were recorded in 10 (55%) dogs.
Lower urinary tract disease or urinary catheter placement was in the history of
6 (33%) dogs. Other urinary abnormalities include urethral tear during a
cystoscopy, intermittent urinary catheterization, cystotomy, and obstructive
prostatic cyst. One dog had been diagnosed with diabetes mellitus. On the other
side, four Candida isolates were reported: C. albicans, C. glabrata,
C. tropicalis, and C. parapsilosis in eight cats had candiduria. The
risk factors comprised other lower urinary tract disorders, urethral avulsion
secondary to trauma, perineal urethrostomy, and bladder rupture secondary to
urethral obstruction. Four cats had received immunosuppressive medications.
Administration of antibacterial drugs in the last 30 days was reported for 7 of
the 8 cats (Reagan et al., 2019).
In April
2011, a 3-year-old male Yorkshire terrier dog was referred to the Clinical
Veterinary Hospital de Madrid, Spain, with a diagnosis of relapsing UTI. The
dog had a history of ammonium urate bladder stones and had been treated
previously with marbofloxacin. Microscopic examination and culturing of urine
specimens revealed the presence of yeasts. Yeast isolates recovered from
clinical specimens were identified as Candida tropicalis on the basis of
the morphology and pigmentation of their colonies on ChromAgar medium and also
by sequencing the D1/D2 domains of the large subunit (LSU) rRNA gene
(Álvarez-Pérez et al., 2016).
3.2.
Other Fungal Species
In
October 2020, a 10-year-old male intact Border Collie dog presented to a
specialty veterinary hospital at Michigan State University for a two-month
history mural ureteral and bladder granuloma. The diagnosis, culture followed
by MALDIToF, PCR, and sequencing was performed and identified Scedosporium
apiospermum which is an opportunistic mold that is an emerging disease in
humans and animals (Tsoi et al., 2021). A nine-year-old female Labrador
retriever suffered from urinary tract infection and was in the 14th
day of a 21-day course of oral antibiotics amoxicillin-clavulanic acid in
Sydney, Australia, in March 2017. The signs were hematuria, sanguinous vulval
discharge, and urinary incontinence. The main compliance was the
ultrasonographic observation of two intra-abdominal masses, "eumycetomas"
which are chronic pyogranulomatous lesions caused by molds. Antifungal therapy
was started on day 3 with oral itraconazole at 5mg/kg SID for 90 days. However,
the dog continued to have urinary tract infections and urinary incontinence for
the whole ninety days after treatment commenced. Due to cost constraints,
euthanasia was elected on day 97. Amplification
and sequence analysis of internal transcribed spacers and the partial large
subunit of the 25–28s ribosomal RNA regions of fungus cultured was performed,
identifying this as belonging to the Curvularia species (Herbert et
al., 2019). The Blastomyces spp. organism was detected in urine
sediment obtained from a 2-year-old castrated male Doberman pinscher (Reagan et
al., 2019). A 2.5-year-old female spayed GSD dog was presented to the
University Veterinary Teaching Hospital, Sydney, Australia, for investigation
of polyuria, polydipsia, and urinary incontinence at night of at least four
months duration. Fungal colonies were grown from both urine and lymph node
aspirates. Molecular identification targeting the partial beta-tubulin gene
revealed these colonies to be Aspergillus deflectus (Bennett et al.,
2018). Cryptococcal UTI was diagnosed cytologically and via fungal culture in a
male domestic shorthaired cat with stranguria and pollakiuria.
4.
VIRAL URINARY TRACT
INFECTION
Viral-induced
disease is recognized, particularly of the upper urinary tract. However, it can
be difficult to determine cause-and-effect relationships because viral-induced
illness may occur in the absence of detectable replicating virus. Several
viruses have been implicated in canine and feline disease, as shown in table(1)
by Olin and Bartges, (2015).
Table 1: Viruses associated with urinary tract disease
in dogs and cats. (Olin and Bartges, 2015).
|
Species |
Upper Urinary Tract Disease |
Lower Urinary Tract Disease |
|
Canine |
Canine adenovirus type I |
|
|
Canine herpesvirus |
|
|
|
|
|
|
|
Feline |
Feline coronavirus |
Feline calicivirus |
|
Feline immunodeficiency
virus |
Bovine herpesvirus-4 |
|
|
Feline leukemia virus |
|
|
|
Feline foamy (syncytium-forming) virus |
Feline foamy (syncytium-forming) virus |
DIAGNOSIS
In 2019,
the International Society for Companion Animal Infectious Diseases (ISCAID)
released revised guidelines for the diagnosis and treatment of bacterial UTIs
in pets. The recommended diagnostic method for UTIs combines clinical signs,
urinalysis; including dipstick testing, specific gravity measurement, and
sediment cytology, haemato-biochemical analysis, radiography, and
ultrasonography as well as bacterial culture results obtained from urine
samples collected using cystocentesis (Barot et al., 2022).
4.1.
Clinical examination
Clinical signs associated with UTI are variable and
depend on the interaction of (1) virulence and numbers of the uropathogen, (2)
presence or absence of predisposing causes, (3) the body's compensatory
response to infection, (4) duration of infection, and (5) site (s) of
infection. Pollakiuria, stranguria, dysuria, hematuria and inappropriate
urination may be observed with lower UTI (Bartges, 2004).
4.2.
Sampling
Free-catch urine sampling
When
collecting a free-catch urine sample, a mid-stream sample is preferred. The
initial urine that is voided contain cells, bacteria, and debris from the
urethra and vulva or prepuce, and may not be a representative sample. Because
female dogs are usually positioned with the vulva close to the ground when
urinating, a shallow container (such as a pie pan) may be helpful. Quantitative
analysis should be performed on all free-catch samples submitted for bacterial
culture.
Obtaining
a free-catch midstream urine sample from a cat is a difficult task. Removing
the litter from the litter box or replacing it with nonabsorbent material,
plastic packing material, etc. may allow collection of a suitable sample.
Plastic wrap can be loosely placed over the litter to collect urine from
declawed cats. Generally, the use of voided urine specimens for bacteriological culture
is discouraged because contamination from external genitalia led to
misinterpretation of laboratory results (Olin and Bartges
2015).
A
study addressed the usefulness of voided specimens and determined veterinary
cut-off values for significant bacteriuria. The
results showed false negative culture results. False negative results
are concerning, as they led to under-treatment, thereby posing a risk of
complications to individual dogs. The veterinary
cut-off yielded an accuracy of 95% with a sensitivity and specificity of 94%. (Sørensen et al., 2016).
Urinary catheterization
Male dogs are placed in lateral or sternal
recumbency for catheterization. The penis is extruded from the prepuce, and the
tip is cleaned with a dilute disinfectant solution to remove any debris or
discharge. Sterile lubricant is placed on the tip of the urinary catheter,
which is then placed in the urethral orifice, and the catheter is advanced into
the bladder (Fig. 4). It is helpful to create a sleeve from the catheter
packaging to maintain sterility while passing the catheter. Most female dogs need chemical restraint,
such as lidocaine gel, to allow urinary catheterization (Figure 4).
Both male and female cats usually need
chemical restraint to permit urinary catheterization. Generally, it is not
routinely used for urine collection in cats unless it has been performed for
another reason such as treating urethral obstructions (Reppas and Foster, 2016).
After passing a urinary catheter to obtain a urine sample, the first few
milliliters of urine should be discarded, as that is the portion most likely to
have debris from the urethra. A second aliquot can then be obtained for
urinalysis and quantitative culture if desired.
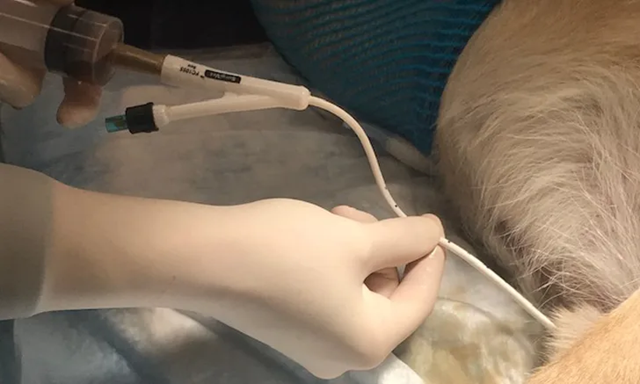
Figure 4: Urethral
catheterization placement in male dog.
https://www.cliniciansbrief.com/article/urinary-catheter-placement-dogs
Cystocentesis
Cystocentesis allows the collection of an
uncontaminated urine sample ideal for bacterial or fungal culture and can be
performed on awake animals. It also allows appropriate timing of collection
when timing is important.
The dog is usually positioned in dorsal
recumbence. Palpating the bladder with one hand helps immobilize it while
acquiring the sample. The needle should be angled caudally, toward the pelvic
inlet, so that as the bladder empties, the needle is still in the lumen of the
bladder (Fig. 5). It is also possible to use a lateral or standing approach
when the bladder is palpable. A 1-inch, 22-gauge needle is preferred, and for
very large or obese dogs, a 1.5 to 2-inch needle may be required.
Figure 5: Cystocentesis (ie, obtaining urine directly
from the urinary bladder by inserting a needle through abdominal wall).
https://www.cliniciansbrief.com/article/cystocentesis
In cats, cystocentesis is usually performed
relatively easily unless they have idiopathic feline lower urinary tract
disease (iFLUTD). These cats typically have a small bladder, and any handling
results in voiding; ultrasound-guided sampling may be more rewarding in these situations (Reppas and Foster, 2016).
Notably, with both cystocentesis and urinary
catheterization, there is a risk of iatrogenic hematuria. Iatrogenic hematuria
can occur if the needle is in contact with the opposite bladder wall. Other
complications include puncture of the colon, laceration of the bladder, and
laceration of the major blood vessels dorsal to the bladder. Inadvertent
puncture of the colon caused bacterial contamination of the urine sample, and a
mixed population of bacteria on the urine culture (Esparaz et al., 2016).
Storage of
samples
The storage condition was vital to
maintaining the quality and accuracy of the urine samples. A study assessed
different storage conditions of urine samples obtained from 30 dogs and 49
cats. The study showed that storing conditions at room temperature or
refrigeration for 24 h do not impact the results of culture count in cat urine
samples. For dog samples, chilled samples have a higher accuracy rate than room
temperature samples, although the overall agreement was still satisfactory (Lien and Wang, 2023).
Additionally, Hedström, et al. (2021) declared that boric acid sponge
preservation is a useful alternative to refrigeration of urine samples during
transport. Reliable quantitative bacterial culture results can be obtained from
canine urine up to 48 h after collection if urine is refrigerated and for at
least 24 h if urine is stored using a boric acid-containing urine transport
system.
4.3.
Urinalysis
A urinalysis should be performed routinely as
part of a minimum database and is an essential part of the diagnostic
evaluation for all urinary and many metabolic diseases (Bartges, 2004).
A complete urinalysis assessment includes
determining urine specific gravity (USPG) using a refractometer, evaluation of
physical characteristics (color, clarity, and volume), biochemical parameters
(urine pH, blood, glucose, ketones, bilirubin, urobilinogen, and protein) using
analytic test pads on dipsticks, and microscopic sediment evaluation (RBC, WBC,
organisms, epithelial cells, crystals, and casts) (figure 6). Collection of
urine by cystocentesis is the preferred method when evaluating patients for UTI.
If infectious prostatitis or vaginitis is suspected, different techniques were
indicated (Reine and Langston, 2005).

Figure (6): Normal values,
urinalysis in dog and cat (Bartges, 2004).
The physical
characteristics of urine should be inspected when conducting urinalysis on
every sample. These features are easily observed by both client and clinician
alike and, as such, add no expense to case management. Urine volume, color,
clarity, and odor are often overlooked. The veterinary team would do well to
reconsider these urine properties because they provide unique insight about pet
health and ongoing disease processes. Physical properties of urine also prompt
pattern recognition. For example, observing turbid urine in canine or feline
cases generates a list of the most common differentials, including pyuria,
crystalluria, and bacteriuria. These clues guide decision-making as clinicians
determine what is most likely ongoing in the affected case and how best to
achieve a definitive diagnosis (Englar, 2022).
The dipstick was proven
useful for rapid urinalysis to evaluate urine specific gravity (USG), pH,
leukocytes, nitrites, glucose, proteins, ketones, urobilinogen, bilirubin, and
blood. The USG and pH significantly changed during the neonatal period. Other
parameters did not vary significantly in relation to age (Melandri et al.,
2020).
Dipstick urinalysis is
easily performable under every condition and is a cheap and repeatable
diagnostic test, providing immediate results. A drop of urine can be put on
each field of the strip to obtain reliable results using small volumes of
urine. After dripping, the results were read after 60 seconds for all the
parameters, except for leukocytes that were read at 120 seconds (Balogh et
al., 2017).
Urinary pH is linked to
alimentary habits; in carnivores, including dogs, it normally ranges between 6
and 6.5, being slightly acidic, and the urinary pH is influenced by alterations
in systemic homeostasis.
Sediment evaluation
Normal urine should contain very
few red blood cells. Owners may observe macroscopic hematuria, but microscopic
hematuria will go undetected without sediment evaluation. Hematuria resulted
from pyelonephritis, bladder infection, genitourinary tract inflammation,
neoplasia, bleeding disorders, or trauma (McGuire et al., 2002). The
existence of increased WBC indicated the existence of urinary tract
inflammation, typically greater than 5 WBC/high power field (HPF). The presence
of pyuria certainly raises concern about the presence of a bacterial urinary
tract infection. Pyuria in a sample obtained via cystocentesis indicated
infection or inflammation of the kidneys, ureters, urinary bladder, or
prostate. Any case with pyuria, particularly those with signs referable to the
urinary tract (i.e., stranguria, pollakiuria, hematuria, polyuria), should have
a urine culture and sensitivity performed. Dogs with diabetes mellitus are
predisposed to urinary tract infection and many times do not have significant
pyuria (McGuire et al.,
2002).
Normal urine is sterile. Because
bacteria can be found in the distal urethra and genital tract, samples obtained
via free catch or catheterization may have some degree of contamination.
Cystocentesis is the method of choice for obtaining the urine sample when
bacterial urinary tract infection is suspected. Although bacteria can be
visualized on microscopic examination, it is sometimes difficult to distinguish
bacteria from debris. The presence of pyuria in the same sample would support
the finding of bacteriuria. Microscopic examination of modified Wright’s
stained urine samples has been shown to be superior to traditional wet mounts
when attempting to identify bacterial urinary infections in dogs. A bacterial
culture and sensitivity should be performed on urine samples with microscopic
evidence of bacteriuria (Swenson et al., 2004).
Occasionally, yeast or fungal hyphae
may be seen in a urine sample and often represent contamination. In cats, true
fungal urinary tract infections are most commonly seen associated with
prolonged antibiotic and/or glucocorticoid therapy, aciduria, and the use of
indwelling transurethral catheters. Fungal organisms can also be identified in
the urinary bladder of pets with systemic mycoses (i.e., blastomycosis) (Werner and Norton, 2011).
Increased
numbers of epithelial cells can be seen in association with infection,
inflammation, irritation, and neoplasia. It is regarded as the gold standard method to
attain an accurate diagnosis of bacteriuria and to gain the best course of
treatment based on a decision-making process to limit the spread of resistance.
Importantly, cultures should be repeated three to five days after the
termination of antimicrobial therapy to ensure elimination of infection (Rampacci et al.,
2018).
The quantitative bacterial culture (QBC)
using the Calibrated Loop/Surface Streak Method (Fig. 7) is considered the gold
standard for determining UTIs before initiation of antimicrobial therapy. It is
necessary for differentiation of harmless bacterial contaminants from bacterial
pathogens that facilitating accurate identification of specific bacterial species
aids in selection of antimicrobial drugs. It also facilitates differentiation
of recurrent UTIs caused by relapses from recurrent UTIs caused by reinfections (Strachan et al.,
2022). One µL of urine sample was inoculated using a sterile
inoculating loop at the surface of the standard nutrient agar plate with a
single streak across the center. Then, spread the inoculum in a cross-zigzag
manner (Fig. 7). After overnight incubation at 37°C, colonies were counted, and
the number of colony-forming units (CFUs) per milliliter of urine was
calculated. A quantitative urine culture demonstrated significant bacterial
infection in the case of ≥ 103 CFU/ml in cystocentesis samples (Karah et al., 2020).

Figure 7. Urine culture using the
calibrated loop/surface streak method.
The general-purpose media are sufficient for
urine culture in low-resource settings; according to traditional guidelines,
blood agar (non-selective medium) and MacConkey agar (selective and
differential for Gram-negative rods) are probably the most commonly recommended
and used media for routine urine cultures. As an alternative, cysteine lactose
electrolyte-deficient (CLED) agar or chromogenic agar has been proposed as
standard media for urine culture. Eosin methylene blue agar (EMB) was used as a
differential for suspected E. coli colonies that were subcultured with the
formation of a characteristic green metallic sheen. Mannitol salt agar is used
as a selective medium for the isolation of Staphylococcus spp. Sabouraud agar should be added, in
addition to the usual bacterial media, to culture the urine sample in
particular care units or if yeasts have been seen by microscopic examination.
The choice of media for routine urine culture should be made locally based on
available resources and the desired approach of identification. Blood agar is
replaced by nutrient agar in order to keep the costs low and since
Gram-negatives have frequently accounted for the majority of anticipated
pathogens. Isolates were identified based on colony morphology and characteristics (Public Health, 2020).
In freshly voided urine, the culture of ≥ 100
colonies of one type (number of bacteria is ≥ 105 cfu/mL) has usually been
regarded as a cutoff for UTI. If 10–100 colonies of one type are counted
(number of bacteria is between 104 and 105 cfu/mL), the result should be
evaluated according to the clinical status. On the other hand, the probability
of UTI is low if the number of colonies is <10 (number of bacteria is <
104 cfu/mL). For cultures containing two types of colonies, UTI is likely if
≥100 colonies are counted for at least one of the two types. Subcultures, for
further identification and antimicrobial susceptibility testing, should be
performed for each type counting ≥ 100 colonies, but it is also recommended to
request a new sample. Notably, if both types have <100 colonies, for each
one, UTI is not likely, and the sample is often contaminated. If there are more
than two types of colonies, the sample is often contaminated. A new sample
should be requested (CDC, 2020).
The sensitivity and specificity of Gram stain
were evaluated on 103 canine urine samples acquired via cystocentesis and
suspected UTIs. The centrifuged urine from the urine sediment preparation was
used to make slides for Gram staining. These slides were allowed to air dry and
then heat-fixed and stained with a commercially available Gram stain kit. A
quantitative assessment of bacteria (none <3, 3–6, 6–10, 10–20, 20–50,
50–100, 100–200, 200–500, >500 bacteria/HPF) was recorded for each sample.
Any slide with bacteria was subsequently assessed for bacterial morphology (rod
or cocci) and whether these bacteria stained Gram positive or Gram negative.
Slides were considered positive for bacteriuria if ≥1 bacteria per HPF was
observed and parallel to positive urine culture. The results revealed that Gram
stain demonstrated 96% sensitivity, 100% specificity, 100% positive predictive
values (PPV), and 93% negative predictive values (NPV) in the detection of
bacteriuria for all dogs. Gram staining should be considered when bacteriuria
is highly suspected and requires rapid identification while bacterial culture
is pending (Way et al., 2013).
A number of 459 urine samples collected by
cystocentesis from dogs suffered from UTIs; signs were prepared as unstained
wet-mounted air-dried urine sediment. The preparations were subjected directly
to modified Wright stain and examined for the presence of bacteria. Compared
with the results of quantitative bacteriologic culture, modified Wright-stained
preparations had a sensitivity of 93.2%, a specificity of 99.0%, PPV of 94.5%,
NPV of 98.7%, and a test efficiency of 98.0%. So examination of urine sediment
preparations by modified Wright-stained appeared to be a rapid, cost-effective
method that significantly improved the sensitivity, specificity, PPV, NPV, and
test efficiency of light microscopic detection of bacteriuria (Swenson et al.,
2004).
4.4.
Biochemical
Characterization
Positive urine culture is usually followed by
a variety of biochemical identification tests to determine the species/genus of
the implicated bacterium. The identification of bacterial pathogens is divided
into two levels The Basic (level 1)” shall be available one day after receiving
the sample and the advanced (level 2).” shall be ready in two days after
receiving the sample (Table 2&3) (Karah et al., 2020).
Table 2.
Basic biochemical identification of common uropathogens. (Karah et al.,
2020).
|
Bacterium |
Mac 1 |
Gram Stained Bacterial Cell Morphology |
Glu 1 |
Oxi 1 |
Cat 1 |
PYR 1 |
Lanc 1 |
|
Enterobacterales |
+ |
Red or pink rod-shaped |
+ |
– |
NA |
NA |
NA |
|
Pseudomonas-like glucose-non-fermenter
Gram-negative rods |
+ |
Red or pink rod-shaped |
– |
+ |
NA |
NA |
NA |
|
Acinetobacter-like
glucose-non-fermenter Gram-negative rods |
+ |
Red or pink rod-shaped |
– |
– |
NA |
NA |
NA |
|
Staphylococci |
– |
Clusters of purple or mauve sphere-shaped |
NA |
NA |
+ |
NA |
NA |
|
Enterococci |
– |
Pairs or short chains of purple sphere-shaped |
NA |
NA |
– |
+ |
D |
|
Streptococci |
– |
Chains of purple or mauve sphere-shaped |
NA |
NA |
– |
– * |
B or D |
Mac,
MacConkey Agar; Glu, glucose fermentation; Oxi, oxidase; Cat, catalase; PYR,
Pyrrolidone arylamidase; Lanc, Lancefield grouping; NA, Not Applicable. *
Except for streptococci group A, which is not a common uropathogen.
Table 3. Advanced biochemical identification of common
uropathogens (Karah et al., 2020).
|
Enterobacterales |
Lac 1 |
Ind 1 |
Cit 1 |
VP 1 |
Ure 1 |
Mot 1 |
H2S 1 |
LDC 1 |
Nit 1 |
|
|
Escherichia coli |
+ |
+ |
– |
– |
– |
+ |
– |
+ |
+ |
|
|
Klebsiella pneumoniae |
+ |
– |
+ |
+ |
+ |
– |
– |
+ |
+ |
|
|
Klebsiella oxytoca |
+ |
+ |
+ |
+ |
+ |
– |
– |
+ |
+ |
|
|
Enterobacter cloacae |
+ |
– |
+ |
+ |
V |
+ |
– |
– |
+ |
|
|
Enterobacter aerogenes |
+ |
– |
+ |
+ |
– |
+ |
– |
+ |
+ |
|
|
Citrobacter freundii |
V |
– |
+ |
– |
V |
+ |
(+) |
– |
+ |
|
|
Citrobacter koseri |
V |
+ |
+ |
– |
V |
+ |
– |
– |
+ |
|
|
Proteus mirabilis |
– |
– |
V |
V |
+ |
+ |
+ |
– |
+ |
|
|
Proteus vulgaris |
– |
+ |
(–) |
– |
+ |
+ |
+ |
– |
+ |
|
|
Providencia stuartii |
– |
+ |
+ |
– |
V |
(+) |
– |
– |
+ |
|
|
Morganella morganii |
– |
+ |
– |
– |
+ |
+ |
– |
– |
+ |
|
|
Serratia marcescens |
– |
– |
+ |
+ |
(–) |
+ |
– |
+ |
+ |
|
|
Glucose-non- |
Oxi 1 |
Lac 1 |
Ind 1 |
Cit 1 |
VP 1 |
Ure 1 |
Mot 1 |
H2S 1 |
LDC 1 |
Nit 1 |
|
P. aeruginosa |
+ |
- |
- |
V |
- |
(-) |
+ |
- |
- |
V |
|
A. baumannii |
- |
- |
- |
+ |
- |
- |
- |
- |
- |
- |
|
Staphylococci (catalase-positive) |
Slide Agg 1 |
Tube Coag 1 |
Hemolysis |
Salt Tol 1 |
Mann 1 |
Nov 1 |
||||
|
Staphylococcus aureus |
+ |
+ |
V |
+ |
+ |
S |
||||
|
S. saprophyticus |
- |
- |
None * |
+ |
+ * or - |
R |
||||
|
S. epidermidis group |
- |
- |
None * |
+ |
- |
S |
||||
|
Streptococci
(catalase-negative) |
Lanc 1 |
Hemolysis |
Bile esc 1 |
6.5% NaCl tol 1 |
||||||
|
Enterococci |
D |
No hemolysis * |
+ |
+ |
||||||
|
Group D streptococci other
than enterococci |
D |
β-, α-, or no hemolysis |
+ |
- |
||||||
|
Streptococcus agalactiae |
B |
β-hemolysis * |
- |
- |
||||||
Lac, lactose fermentation; Ind, indole; Cit, citrate
utilization; VP, Voges-Proskauer; Ure, urease; Mot, motility; H2S,
hydrogen sulfide production; LDC, lysine decarboxylase; Nit, nitrates
reduction; Oxi, oxidase; Agg, agglutination; Coag, coagulase; Tol, tolerance;
Mann, mannitol fermentation; Nov, novobiocin susceptibility; Bile esc, bile
esculin hydrolysis; +, 90–100% Positive; -, 0–10% positive; (+), 76–89%
positive; (-), 11–25% positive; V, variable.* Most strains.
4.5.
Serological Identification
Smith et al.
(2020a) evaluated urine myeloperoxidase (uMPO) as a
rapidly available, accurate marker to predict urine culture results. They
hypothesized that uMPO would be higher in dogs with a positive urine culture
than in dogs with a negative urine culture and that uMPO could be used to aid
in the accurate diagnosis of significant bacteriuria. The authors measured uMPO
using a commercially available canine myeloperoxidase ELISA on urine samples
from 98 dogs (forty-seven dogs had a negative urine culture and fifty-one dogs
had a positive urine culture). The given results indicated that uMPO levels
were significantly higher in samples that had a positive culture (median 2.13
ng/ml) versus samples that had a negative culture (median 1.07 ng/ml) (p <
0.005). Using a cutoff of 0.55 ng/ml, uMPO had a sensitivity of 70% and
specificity of 69% to determine the presence of a positive culture.
A rapid immunoassay (RIA; BacVet) test
utilizes a cocktail of monoclonal antibodies targeting a panel of bacterial
surface proteins. It was carried out on twenty-one freely voided urine
specimens obtained from dogs, which showed many signs of LUTIs according to the
manufacturer's guidelines in the USA. The sensitivity of the RIA was 89%,
specificity 100%, PPV 100%, and NPV 92%. The study revealed that the simple
point‐of‐care RIA test can be performed in‐office, rapidly, at low‐cost, and
without specialized training (Grant
et al., 2021).
Thereafter,
Grant et al. (2023) evaluated the previous rapid
immunoassay (RIA: BacVet) for the diagnostic performance immediately after
urine collection and after refrigeration at 4 and 24 hours. The study was
conducted on 40 voided urine samples from dogs with clinical signs of LUTIs.
The results showed sensitivity, specificity, PPV, and NPV of the RIA were 100%,
88%, 82%, and 100%, respectively, and results were not different after 4 and 24
hours of refrigeration. The test is inexpensive, rapid, and accurate.
Similarly, in an Australian study, a rapid immunoassay (RIA; RapidBac) was
performed on forty-four urine specimens obtained by cystocentesis from 44 dogs
according to the manufacturer's guidelines for diagnosis of bacteriuria. The
results determined good sensitivity and excellent specificity, 81.8% and 95.5%,
respectively, compared to urine culture (Sutter et al., 2023).
Microscopic
Agglutination Test is the recommended technique for leptospirosis diagnosis, as reactivity
to a serovar indicates exposure to a corresponding serogroup. Antibodies (IgM
and IgG) were detected around 15 days post-infection, but little information
was available on their duration in pets' blood. Clinical interpretation relies
on paired serum titres, and some infected animals may produce lower results
than the accepted 1:100. It is even possible that seroconversion in cats was
expressed at a lower titre compared with dogs (Shropshire et al.,
2016).
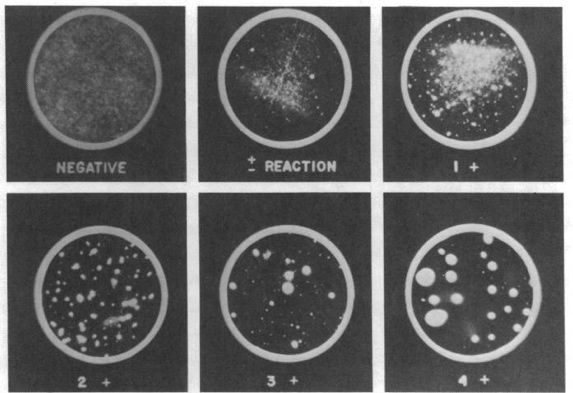 The serum of 112 cats was investigated by
MAT, detecting anti-Leptospira antibodies against 14 pathogenic serovars.
Antibodies against 6 serovars—Poi, Bratislava, Arborea, Ballum, Pomona, and
Lora—were detected in 15.3% (17/111) of cats (titers range: 20-320) as shown in
figure (8) (Donato et al., 2022). A study
in Indian Ocean islands addressed stray and domestic cats (n = 92) using a
serological MAT. The results revealed a seroprevalence of 37.0% (34/92)
(cut-off 1:40) without a significant difference in the living conditions of
animals. The predominant serogroup was Icterohaemorrhagiae, but Ballum,
Cynopteri, and Australis were also detected (Holzapfel et al.,
2021).
The serum of 112 cats was investigated by
MAT, detecting anti-Leptospira antibodies against 14 pathogenic serovars.
Antibodies against 6 serovars—Poi, Bratislava, Arborea, Ballum, Pomona, and
Lora—were detected in 15.3% (17/111) of cats (titers range: 20-320) as shown in
figure (8) (Donato et al., 2022). A study
in Indian Ocean islands addressed stray and domestic cats (n = 92) using a
serological MAT. The results revealed a seroprevalence of 37.0% (34/92)
(cut-off 1:40) without a significant difference in the living conditions of
animals. The predominant serogroup was Icterohaemorrhagiae, but Ballum,
Cynopteri, and Australis were also detected (Holzapfel et al.,
2021).
Figure (8): Microscopic Agglutination Test (MAT) (Donato et
al., 2022).
Delaude et al.
(2017) tested canine urine samples by MAT for antibodies against a panel of 12
serovars. Seropositivity (MAT ≥1:100) was most common to serovars Australis
(14.9%) and Bratislava (8.8%), followed by Copenhageni (6.1%), Canicola (5%),
Grippotyphosa (4.5%), Pomona (4%), Autumnalis (2.7%), and Icterohaemorrhagiae
(1.6%).
Microscopic
Agglutination Test (MAT) test interpretation may be more reliable in cats than in dogs because
no interference with vaccine antibodies exists as cats are not vaccinated.
Furthermore, laboratory-reared young adult specific pathogen-free cats infected
with Borrelia burgdorferi did not form antibodies against Leptospira
species as a cross-reaction. The authors of that study suggest that positive Leptospira
species MAT results from cats in the field are likely to reflect antibodies
against Leptospires and not B. burgdorferi (Murillo et al.,
2020).
ELISA used for leptospirosis identified the
presence of specific IgM leptospiral antibodies earlier than MAT, 4–6 days
post-infection. The main advantages of ELISA compared with MAT are the
stability of antigenic preparations and the genus specificity, meaning all
types of Leptospires can be diagnosed with a single antigenic
preparation, irrespective of the causal serovar. In dogs, a combination of
ELISA and MAT were recommended for leptospirosis diagnosis (Murillo et al.,
2020). An IgM immunoblot assay was 88% sensitive in the first 3
days of human leptospirosis compared with 2% for the MAT. Use of rapid, broadly
reactive antibody assays as screening tests before performing the more specific
and cumbersome MAT may help decrease false negative test results relating to
inadequate serovar inclusion in the MAT. Ideally, they should detect antibodies
that react only with pathogenic serovars. Recombinant LipL32-based assays
recently were evaluated and was found to be sensitive and specific in dogs and
humans compared with MAT testing (Sykes et al., 2011). Rapid
patient-side tests for leptospirosis diagnosis were developed. Curtis et al. (2015) performed a recombinant LipL32-based rapid
in-clinic ELISA (SNAP Lepto) for the detection of antibodies against Leptospira
species in dogs. Neither of the tests distinguishes between serovars, nor do
they provide a titre magnitude. The first test, 66, is based on the detection
of Leptospira-specific IgM and has demonstrated a sensitivity and
specificity of 100% and 95.3%, respectively. It can therefore detect dogs with
clinically suspected acute leptospirosis. Dogs previously vaccinated or
suffering from an acute but subclinical infection can also produce positive
results. A LipL32-based in-clinic ELISA for the rapid detection of Leptospira
specific antibodies in dogs is not IgM specific, but the study authors
considered it a convenient tool to assess Leptospira antibody status in
dogs. Neither rapid test techniques nor ELISA to diagnose leptospirosis in cats
have yet been developed.
4.6.
Molecular
Identification
Generally, the significance of
molecular-based techniques is relied on that they elucidate a reliable, quick,
precise, and low-cost diagnosis, especially in non-culturable or fastidious pathogens.
Melgarejo (2021) used next-generation
sequencing (NGS) and stated that the diversity and abundance of bacterial and
fungal communities varied between urine samples from different dogs.
Pulsed-field gel electrophoresis (PFGE)
identified 31 PFGE patterns among the 43 E. coli isolates, which could be classified into
nine and four groups showing > 80 % and > 95 % similarity. Homology
ranged from 25% to 100%, indicating that canine urinary E. coli isolates exhibited a high degree of genetic
polymorphism (Yu et al., 2020).
A quantitative real-time PCR assay was
developed for the diagnosis and monitoring of mycoplasma UTIs in an English
Cocker Spaniel dog. Attempts to culture organisms from purulent urine failed,
and empirical antibiotic therapy did not resolve the pyuria. A mycoplasma
species most closely resembling Ureaplasma
canigenitalium was
identified in urine samples by conventional PCR and sequencing. The qPCR method
has provided rapid results and achieved verified successful treatment (Hemmatzadeh et al.,
2019). In Leptospira diagnosis, PCR-based techniques
played a very crucial role for detection; partial rrs gene (16S rRNA)
sequencing identified Leptospira
interrogans in all
PCR-positive samples, and Leptospira DNA was detected in renal tissue, urine,
and blood (Holzapfel et al., 2021). A real-time
polymerase chain reaction targeting the lipL32 gene of pathogenic Leptospira
was conducted on feline blood and urine samples. Leptospira spp. DNA was
found in 3% (4/109) of blood and 9% (10/111) of urine samples (Donato et al., 2022).
Positive qPCR canine samples were subjected
to 16S rRNA and secY gene phylogenetic analysis. The recovered strains were
characterized by multilocus sequence typing. Phylogenetic analysis revealed
that 10 dogs had L.
interrogans infection.
Three dogs (3/13) had L.
santarosai infection. The
secY phylogenetic analysis revealed that the L.
santarosai sequences
clustered separately from those obtained from other hosts. The results
suggested a genetic distinction between lineages of Brazilian L. santarosai maintained by dogs and other animal hosts (Miotto et al., 2018).
PCR directly identifies leptospiral DNA. It
does not determine the infecting serogroup or serovar, but it can indicate the Leptospira species. The test can be performed on
blood, urine, cerebrospinal fluid, and body tissues. In cases of acute
leptospirosis, this would be the test of choice to perform on blood and urine
in cats. Compared with culture, PCR gives fast results, contributing to an
early diagnosis. Real-time PCR techniques were recommended due to their greater
sensitivity and specificity. Genes that have more than one copy in the genome,
such as lig or rrs, were selected for increasing the sensitivity of the
technique. Genes present only in the pathogenic species can also be added, as
they will increase the specificity of the test. A positive PCR result means
that leptospiral DNA is present in the sample. In acute infections or in
chronic carriers, the test would be positive in urine, indicating that
bacterial DNA is being shed. However, negative results in blood and urine do
not rule out leptospirosis, as leptospiraemia is transient (only occurring in
the initial phases of the disease); also, results are usually negative if the
pet has received antibiotic therapy, and shedding in urine can be intermittent (Dorsch et al., 2017).
MicroRNAs (miRNAs), classified as non-coding RNAs, regulate various metabolic
systems and viral life cycles. Feline foamy virus (FFV) was identified in
feline urine samples using expression of miRNAs and confirmed by application of
dual-luciferase reporter assay. It was found that the seed sequences of the
miRNAs identified in the study were conserved among all previously reported FFV
isolates. These obtained results suggest that FFV-derived miRNAs played a
pivotal role in FFV infection (Aso et al., 2021).
4.7.
Ultrasonography for
pet animals
It is a significant diagnostic tool and an
ideal noninvasive approach to evaluate urinary tract disorders because it is
easy to implicate, low cost, and provides high real-time contrast resolution.
Ultrasonography is commonly utilized as the first diagnostic technique in
instances of hematuria or dysuria (Fig. 9), as it enhances the diagnosis of
cystolith, nephroliths, renal mass, cystitis, and hydronephrosis (Barot et al., 2022).
For ultrasonographic examination, the animals
were restrained and positioned in lateral or dorsal recumbence. The abdomen was
shaved and coated with an ultrasonic coupling gel, then examined with a probe
of 9–3 MHz. Ultrasonography was used to inspect the urinary bladder’s wall
thickness, urine content, and turbidity, or the presence of any abnormalities
such as cystic calculi, polyps, or tumors. The kidneys were examined for the
presence of cysts, abscesses, or any abnormalities, and the male animals were inspected
for any prostatic cyst or abscess to be excluded (Mantis, 2008).
Figure (9): Ultrasonography for
dogs (Barot et al.,
2022).
4.8.
Clinical diagnostic
approaches
Sporadic bacterial cystitis
Sporadic bacterial cystitis is a common
condition in dogs and less encountered in cats, in which a bacterial infection
of the bladder results in inflammation and corresponding clinical symptoms,
which can involve dysuria, pollakiuria, stranguria, hematuria, or a combination
of these symptoms. Previously, ‘simple uncomplicated’ or ‘complicated’ urinary
tract infection (UTI) has been used to describe bacterial cystitis in dogs and
cats (Jessen et al., 2015).
Diagnosis is based on the presence of lower
urinary tract signs, ideally with concurrent evidence supporting bacterial
cystitis (e.g., hematuria, pyuria, cytologically evident bacteriuria) and
bacterial culture results.
Urinalysis (dipstick, urine specific gravity,
and cytological examination of the sediment) should be performed in all cases
to provide supporting evidence and detect potential comorbidities (e.g.,
glucosuria, crystalluria).
Specimens for culture should be collected by
cystocentesis unless there is a contraindication (which would rarely be present
in animals with sporadic cystitis) or significant difficulties in sample
collection are anticipated (e.g., from a large, morbidly obese dog). Ultrasound
guidance facilitates cystocentesis and assess the bladder for abnormalities
such as uroliths or masses (Patterson et al.,
2016).
Culture of voided samples should only be
performed when cystocentesis is contraindicated because of the potential for
both false positive and false negative cultures. Voided samples should only be
cultured if they are refrigerated and processed by the diagnostic laboratory
within a few hours or cultured in-house (Sørensen et al., 2016). The level
of growth (105 CFU/mL), bacterial species (i.e., and whether pure growth is
present) are important factors to assess when evaluating culture results from
voided samples, along with urine cytology and clinical signs.
Recurrent bacterial cystitis
Recurrent bacterial
cystitis definition in veterinary medicine is similar to that in human
medicine. A diagnosis of three or more episodes of clinical bacterial cystitis
in the preceding 12 months or two or more episodes in the preceding 6 months (Arnold
et al., 2016). Recurrent cystitis may result from relapsing or
persistent infection or reinfection. Consideration of which of these is likely
present can be useful for determining the diagnostic plan (e.g., evaluation of
a nidus of infection vs. reasons for susceptibility to repeated infections).
Diagnosis since recurrent cystitis can be associated with ultrasound, plain
radiography, contrast imaging, or possibly cystoscopy, they may be considered
for refractory clinical recurrent cystitis cases to investigate further for
underlying comorbidities and obtain a biopsy of the bladder mucosa, if
clinically indicated. If clinical signs persist despite negative urine
cultures, biopsies of bladder mucosa can be obtained during cystoscopy and
submitted for culture and histological examination to evaluate for deep-seated
bladder infections or other causes.
1) Urine culture, ideally from a sample collected via cystocentesis,
should be performed in all animals with recurrent cystitis.
2) A diagnostic plan should be established for every animal with
recurrent cystitis.
3) If the pathogen isolated from the animal with recurrent infection is
different from previous organisms isolated, reinfection is likely, and efforts
should be undertaken to identify and address any predisposing factors.
Upper urinary tract infections (pyelonephritis)
Pyelonephritis is an infection
of the renal parenchyma that can occur from ascending infection or bacteremia,
often with Enterobacteriaceae causing the majority of infections (Wong et
al., 2015). In human medicine, acute pyelonephritis is classified as
‘uncomplicated’ or ‘complicated’. Uncomplicated implies there is no underlying
comorbidity; complicated suggested the presence of a systemic disease such as
diabetes mellitus or neoplasia or an anatomical/obstructive disorder such as
urinary stone disease or ectopic ureter. Ascending infection can result
from clinically evident lower urinary tract disease. Additionally,
leptospirosis must be considered in endemic regions (Sykes et al., 2011).
A definitive diagnosis is
difficult, and signs attributable to pyelonephritis can be vague. As opposed to
bacterial cystitis, where morbidity is relatively low, pyelonephritis can
result in severe and rapid kidney injury. Thus, rapid diagnosis is important
for proper treatment. Diagnosis of acute pyelonephritis can be suspected based
on positive aerobic bacterial urine culture when accompanied by systemic signs
such as fever, lethargy, and/or polyuria/polydipsia or renal pain on abdominal
palpation. Laboratory findings of azotemia, casts, and peripheral neutrophilia
with or without left shift. However, animals with acute pyelonephritis may be
oliguric or anuric or have vague clinical signs. Imaging findings such as renal
pelvic dilation and/or blunting of the renal papilla on ultrasound examination
may be noted but are nonspecific (D’Anjou et al., 2011).
Increased concentrations
of biomarkers such as serum creatinine or serum symmetric dimethylarginine
(SDMA) can also support the presence of renal injury (Dahlem et al.,
2017) in association with bacteriuria, but are indicators of glomerular
filtration rate and are not specific for bacterial pyelonephritis as the cause
of kidney injury.
o Culture cystocentesis specimens and susceptibility testing should always
be performed.
o Obtaining a urine specimen for cytology and culture by pyelocentesis
should be considered, particularly if results of culture of a cystocentesis
specimen are negative, or when a cystocentesis specimen can not be obtained.
o Blood cultures are recommended at the same time as urine cultures in
immunosuppressed or febrile animals.
o It is important that culture specimen submissions indicate that
pyelonephritis is suspected to ensure that urine breakpoints are not applied.
o If multiple organisms are isolated from urine, the suspected relative
relevance of these should be considered. This assessment would include the
bacterial species and colony counts.
o Evaluation for leptospirosis should be considered in culture-negative
dogs by use of serological testing and PCR (Sykes et al., 2011).
Subclinical bacteriuria
Subclinical bacteriuria is defined as the presence of bacteria in urine
as determined by positive bacterial culture from a properly collected urine
specimen, in the absence of clinical evidence of infectious urinary tract
disease. Terminology such as ‘urinary tract infection’ or ‘occult infections’
has been used in reference to animals with positive bacterial cultures but no
clinical signs of lower urinary tract disease (Peterson et al., 2012);
however, this terminology now should be avoided.
The term bacteriuria has been used to describe cases where bacteria are
visible cytologically, irrespective of culture results (Way et al.,
2013); however, diagnosis of bacteriuria should be based on culture (Nicolle et
al., 2005). Cytological evaluation is an important part of urinalysis in
animals with suspected urinary tract disease. An increased urine sediment white
blood cell count has been associated with increased odds of a positive culture
(O’Neil et al., 2013), but this has not been a consistent finding
(McGuire et al., 2002). Poor agreement between cytological detection of
bacteria and positive urine culture has been reported in dogs (McGhie et al.,
2014). Increased urine sediment red blood cell count is also not predictive of
positive cultures (O’Neil et al., 2013). Thus, cytological data are
useful adjunctive data to assess animals with potential urinary tract disease
but may not be highly predictive of culture results, infectious disease, or
correlate well with clinical signs of upper or lower urinary tract disease.
Similarly, proteinuria is not predictive of subclinical bacteriuria (Lippi et
al., 2022).
Subclinical bacteriuria is common, even in individuals with no known
predisposing factors. Rates of 2.1–12% have been reported in healthy dogs
(McGhie et al., 2014), with higher rates (15–74%) in groups such as dogs
with diabetes mellitus, morbidly obese dogs, puppies with parvoviral enteritis,
dogs with acute disk herniation, chronically paralyzed dogs, and dogs treated
with cyclosporine or glucocorticoids (Baigi et al., 2017). Study of
subclinical bacteriuria has been limited in cats, and the prevalence may be
lower than reported in dogs; however, rates of 1–13% have been reported in
healthy cats (White et al., 2016; Puchot et al., 2017).
No evidence of an association between subclinical bacteriuria and risk
of development of cystitis or other infectious complications has been reported
in dogs or cats (Wan et al., 2014; White et al., 2016).
Culture of urine from animals
with no evidence of urinary tract disease should not be performed when there
would be no indication to treat based on a positive culture result (McGuire et
al., 2002).
A diagnosis of subclinical
bacteriuria is made based on identification of bacteria by culture of urine
collected via cystocentesis in an animal without clinical signs attributable to
bacterial cystitis.
Cystocentesis is the preferred
method for urine collection, and urine should not be collected by other methods
unless there are contraindications to cystocentesis.
Bacterial cell count, typically
expressed as CFU/mL, can not differentiate subclinical bacteriuria from
bacterial cystitis. Subclinical bacteriuria is differentiated from bacterial
cystitis by the absence of clinical signs and not by the bacterial load. There
is no evidence that high CFU counts indicate a greater risk of disease
development. Subclinical bacteriuria is also not defined by the presence or
absence of pyuria on urine sediment examination.
Re-testing of bacteriuric animals
is not recommended.
6.
TREATMENT
Generally according to ISCAID guidelines, UTIs are treated with
antibiotics. Initially, an antibiotic ‘empirical’ may be prescribed that
targets the most likely bacteria causing infection. After the culture finalizes
(which may take a few days), the veterinarian specialist may need to change the
antibiotic if the results indicate the first antibiotic is not ideal.
Antimicrobial therapy is indicated in most cases while awaiting culture and
susceptibility results to relieve patient discomfort. In most situations,
amoxicillin (11–15 mg/kg PO q8h), amoxicillin-clavulanic acid, and
trimethoprim-sulphonamides (15 mg/kg PO q12h) are considered as the first
empirical antimicrobial choices for UTI treatment in pets. Meanwhile, nitrofurantoin,
fluoroquinolones, and 3rd generation cephalosporins are only recommended if
resistance to first-line antimicrobials is detected or the condition of the pet
warrants it (Weese et al., 2019).
The intention of antimicrobial therapy is to eliminate the bacterial
growth in the urinary tract utilizing an antimicrobial agent in a
cost-effective manner. The degree of infection is dependent on the
susceptibility of the bacteria to the concentration of the antimicrobial agent
reached in the urine. Antimicrobial agents can eliminate the bacterial growth
in the urinary tract within an hour. An effective antimicrobial agent generally
attains minimal inhibitory concentration (MIC) both in the serum and urine of
healthy adults.
The urinary levels are frequently manyfold larger than the serum levels.
However, the serum levels are critical in cases with urinary infections.
Traditionally, antimicrobial therapy has been used for the treatment of UTIs
using either a prophylactic or therapeutic approach. Antibiotics such as
penicillins, sulfanilamide, and cephalexin have been used in the RUTI therapy
(Bader et al., 2020). Following the last update, treatment of
subclinical bacteriuria is not indicated for humans, dogs, or cats”. Up to 12%
of healthy dogs and 13% of healthy cats have subclinical bacteriuria. Even if
pyuria is present along with bacteriuria, antibiotic therapy is not recommended
in the absence of clinical signs of cystitis (Acierno et al., 2024).
6.1.
Antimicrobial Susceptibility Test (AST)
The AST is usually done by the Mueller-Hinton broth micro-dilution
method.
Assays were performed according to CLSI guidelines in triplicate, with
the exception of the indicated procedural variations and use of nonstandard AST
media. The experimental parameters are varied one at a time during testing, and
results were compared with those obtained using the standard reference Broth
microdilution (BMD) method or Cation Adjusted Mueller Hinton Broth (CAMHB).
Falcon 96-well polystyrene plates were used, except for indicated analyses
using Falcon 96-well polypropylene plates. Assay plates were incubated for 18 h
at 35 ± 2°C in ambient air, and MICs are determined visually unless otherwise
indicated. MIC50 and MIC90 values were defined as the minimum concentration of
an antimicrobial agent necessary to inhibit the growth of ≥ 50 % and ≥ 90 % of
the isolates, respectively. MIC50 and MIC90 determinations are important for
assessing the efficacy of a given antimicrobial. Reference strains of tested
bacteria were used as quality control strains. Quality control standards and
test results were interpreted with reference to CLSI documents (CLSI, 2023).
6.2.
Clinical Therapeutic Approach
Sporadic bacterial cystitis
Clinical signs are a result of inflammation.
In dogs, a decision to start antimicrobial therapy while awaiting culture
results (if samples are submitted) is reasonable. However, there is evidence
from humans that analgesics alone may be as effective as antimicrobials in uncomplicated
cases (Gágyor et al., 2015; Bleidorn et al., 2016), which
could be applied to sporadic cystitis in cats and dogs. Consideration can be
given to prescribing an initial course of analgesics (e.g., NSAIDs) and adding
antimicrobials 3–4 days later if clinical signs persist or worsen. Regardless,
NSAIDs (use with caution in cats) should be considered during the initial
treatment period to help ameliorate clinical signs. To avoid unnecessary
antimicrobial use in cats, withholding antimicrobial treatment pending the
result of aerobic urine culture is reasonable.
Optimal empirical choices vary based on the
pathogen and resistance patterns in the region. However, amoxicillin is a
reasonable first choice in most areas. If amoxicillin without clavulanic acid
is not readily available, use of amoxicillin/clavulanic acid is reasonable.
Evidence of a need for clavulanic acid is lacking, and it may not be necessary,
even in infections with beta-lactamase-producing bacteria, because of the high
amoxicillin concentrations that are achieved in urine. Trimethoprim-sulfonamides
(trimethoprim-sulfadiazine, trimethoprim-sulfamethoxazole) are other first-tier
options but may be associated with greater adverse effects. However, the
likelihood of adverse effects is low with short courses of therapy as are
recommended below (Weese et al., 2019). The
recommended duration of therapy is 3–5 days. The short end of that dosing
period may be optimal, but veterinary research to support this is currently
limited (ACADI, 2019).
Nitrofurantoin, fluoroquinolones, and 3rd
generation cephalosporins should be reserved for sporadic cystitis where
amoxicillin (±clavulanic acid) and trimethoprim-sulfonamide are not appropriate
based on culture and susceptibility testing results or case factors. These
drugs can be effective but uncommonly needed, and their use in animals is
scrutinized because of concerns regarding antimicrobial resistance and public
health. Rarely, the dosing regimens that some of these drugs offer (e.g., once
daily administration or single injection) may be required for proper treatment,
and owner compliance is an important consideration. However, clinicians must
differentiate between need and convenience when choosing one of these drugs
over recommended first line options. Additionally, the US FDA has discouraged
routine use of fluoroquinolones in humans for uncomplicated infections because
of adverse effects on joint, tendon, and nerve damage (Weese et al., 2022).
It is not necessary to administer fluoroquinolones in most cases of sporadic
cystitis when other alternatives exist. Treatment of intact male dogs with no
evidence of prostatitis, as well as dogs with comorbidities not involving the
urinary tract and with non-recurrent infections, should be approached as described
above, with the understanding the underlying factors might increase the
likelihood of recurrence.
Veterinarians should be aware of local
(ideally clinic-level) antimicrobial susceptibility patterns to help guide
empirical choices. If the expected incidence of treatment failure to a given
antimicrobial increases, an alternate antimicrobial should be considered.
However, care must be taken when interpreting potentially biased data, such as
culture data obtained predominantly from specimens submitted from animals with
refractory or recurrent cystitis. Veterinarians are encouraged to collect
surveillance data on pathogen susceptibility patterns and clinical response to
guide optimal empirical therapy. Consultations with their laboratory microbiologists
are encouraged (Weese et al., 2019).
Infusion of substances (e.g., antimicrobials,
anti-inflammatories, biocides) into the bladder via urinary catheter is not
recommended because of a lack of evidence of efficacy and the potential for
iatrogenic infection, trauma from catheterization, or irritation of the bladder
from infusates. There is currently no evidence that adjunctive treatment
measures (e.g., cranberry extract, D-mannose) are useful for treatment of
sporadic cystitis (Stapleton et al.,
2011).
Follow up
Lack of clinical response within 48 h of
starting appropriate antimicrobials should prompt further investigation to
determine whether cystitis is actually present and identify complicating
factors.
If initial culture results indicate
resistance to the empirical antimicrobial that was chosen, the drug should be
changed unless there has been a good clinical response.
Empirically changing antimicrobials in
response to poor initial response to treatment is not recommended. If clinical
failure has been documented, the cause must be determined, as it may be
unlikely that a different drug will result in a better outcome. Animals with
partial or complete clinical failure to treatment should be re-examined. Unless
the initial culture results indicated resistance to the antimicrobial that was
used empirically, or poor owner compliance is documented, prescribing a new
course of antimicrobials in the absence of further investigation of the reason
for clinical failure is not recommended (Weese et al., 2011).
Post-treatment urinalysis or urine culture is
not recommended for sporadic cystitis when clinical signs have resolved.
Recurrent bacterial
cystitis
Previously, guidelines supported long
durations (4 weeks) of antimicrobials for recurrent cystitis (Weese et al., 2011).
However, recurrent cystitis encompasses a broad range of conditions, including
repeated and relatively uncomplicated infections that likely respond quickly to
antimicrobials and others with marked bladder pathology that complicates
treatment. Broad recommendations for treatment duration are difficult because
of this variation.
The goals of treatment must be considered.
The primary objective is clinical cure with minimal risk of adverse effects
(including antimicrobial resistance). Microbiological cure (elimination of the
offending organism) is desirable but not necessarily achievable or required for
short- or long-term clinical resolution.
Depending on the severity of clinical signs
and the owner’s ability to observe the animal, treatment with analgesics (e.g.,
NSAIDs) alone could be considered while awaiting urine culture results.
However, empirical therapy is reasonable and should be approached as described
for ‘sporadic bacterial cystitis’.
If empirical antimicrobials are initially
prescribed, antimicrobial choice should be reassessed when culture results are
available. If the bacterial strains isolated are reported to be susceptible to
the antimicrobial drug selected, no change in treatment plan is required. If
one or more isolated strains are not susceptible, the animal’s response should
be considered. If clinical cure is documented, it is acceptable to continue
with the initial antimicrobial that was chosen. If clinical failure is documented,
an antimicrobial change is indicated.
Long-term therapy is not automatically
warranted for recurrent cystitis, even in dogs with underlying comorbidities
such as diabetes mellitus, and this is especially true if recurrent disease
appears to be caused by re-infection. Short (3–5 days) durations should be
considered for reinfection. Longer courses (7–14 days duration) may be
reasonable in persistent and potentially relapsing infections if factors that
inhibit response to antimicrobials, such as bladder wall invasion, are
suspected to be present. In those situations, drugs that are ineffective
against Escherichia coli in tissue (e.g., amoxicillin/clavulanic
acid) should be avoided (CLSI, 2023).
Intra-vesicular administration of
antimicrobials or biocides is not recommended.
Follow up
Clinical cure rates are poorly established
for recurrent cystitis. Most monitoring is based on clinical response, as data
on the expected or desired microbiological, cytological, or hematological
response to treatment are lacking.
When short (3–5 days) durations of treatment
are being used, culture during treatment is not recommended. When longer
durations of treatment are being used, the benefit of intra-treatment culture
is unclear (Weese et al., 2011).
When longer durations of treatment are being
used, urine culture is reasonable to consider after 5–7 days of treatment;
however, the approach to a positive or negative result should be considered in
advance. Positive cultures indicate the need for evaluation of compliance and
further diagnostic testing to determine why the bacterium has not been
eliminated, not simply a change in antimicrobial, particularly if clinical cure
has been documented. Negative results could be used to help determine when to
stop therapy if a long course of treatment is being used but are not a
guarantee of microbiological cure.
Culture of urine specimens, ideally collected
by cystocentesis, can be considered 5–7 days after cessation of antimicrobials
in animals where clinical cure is documented. However, this should be used as
part of the diagnostic process to help differentiate relapse, re-infection, and
persistent infection, and to guide potential future diagnostic testing, not as
an indication of a need to treat. The presence of bacteriuria post-treatment
should be approached as described under ‘Subclinical bacteriuria’. If client
compliance is deemed to have been adequate, referral to a specialist should be
considered to explore reasons for microbial persistence or rapid re-infection (CLSI, 2023).
Prevention
Balancing potential efficacy, resistance, and
adverse effects is a challenge, and there are few published studies in dogs or
cats. Single nightly dose nitrofurantoin has been anecdotally used in dogs to
prevent recurrent cystitis, but efficacy data are lacking. Furthermore, adverse
effects of these drugs exist, and there is concern for the selection of
resistant bacteria. Alternative approaches for prevention and treatment of
recurrent cystitis that have been investigated in human beings as well as
animal UTI models include the use of cranberry extract (McMurdo et al.,
2009), cranberry juice (Stapleton et al., 2011), probiotics (Rodrigues et al.,
2014), live biotherapeutic products (such as asymptomatic
strains of E. coli) (Segev et al., 2018),
vaccines (Billips et al., 2009), and various other alternative therapies,
such as methenamine, D-mannose, and intravesicular or orally administered
glycosaminoglycans (Mansour et al., 2014). There has
been limited study in dogs. In one study, cranberry extract prevented adherence
of E. coli colistrains isolated from
dogs to canine kidney cells, and six dogs with recurrent UTI treated with
cranberry extract did not develop UTI when monitored for 2 months (Chou et al., 2016),
but a placebo-treated control population was not studied. In another
prospective, randomized, placebo-controlled study of 94 dogs with thoracolumbar
disk herniation, cranberry extract did not appear to reduce the prevalence of
bacteriuria, with six dogs in the placebo and 11 dogs in the cranberry extract
group developing bacteriuria over a 6-week period (Olby et al., 2017).
Live biotherapeutic products appear promising for treatment of recurrent
cystitis, with a preliminary study reporting complete or nearly complete
clinical cures in four out of nine dogs with recurrent cystitis in response to
instillation of E. coli 2-12 (Segev et al., 2018).
Prophylactic
antimicrobial therapy for dogs and cats is not recommended.
Treatment with a short course (3–5 days
duration) therapy ideally based on susceptibility testing, is most appropriate
to alleviate clinical signs, with a focus on clinical rather than
microbiological cure.
There is insufficient evidence to recommend
the administration of cranberry extract products and other alternative
therapies at this time.
There is insufficient evidence to recommend
administration of methenamine. Data from human medicine suggest it may be
effective in some (but not all) human populations with recurrent cystitis (Lee et al., 2012);
however, evidence of efficacy and safety in dogs and
cats is lacking. Conversion to the active form (formaldehyde) requires low pH,
which is not always assured in dogs and cats with recurrent cystitis.
Upper urinary tract
infections (pyelonephritis)
Treatment should be initiated immediately, while awaiting culture and
susceptibility results.
Initial treatment should involve antimicrobial drugs known to have local
or regional efficacy against Enterobacteriaceae. If regional data are
supportive, a veterinary fluoroquinolone or cefpodoxime are reasonable first
choices. Cefotaxime and ceftazidime are options for IV administration (Table
4).
If ascending infection is suspected, recently obtained urine culture
results should be the basis of initial therapy (remembering that serum
breakpoints must be considered). If hematogenous spread is suspected, initial
therapy should be based on cultures of blood or the infected site whenever
available (CLSI, 2023).
Oral antimicrobial therapy is recommended in animals that otherwise
appear systemically well and have normal appetites. Intravenous therapy is
recommended for animals that are dehydrated, hyporexic, anorexic, or lethargic
as recommended in humans (Strohmeier et al., 2014).
oCulture and susceptibility data should be reviewed when results are
received.
oIf combination therapy was initiated empirically and the isolate is
susceptible to both drugs, one might be discontinued if supported by evidence
of clinical response.
oIf resistance is reported to one of the drugs, that antimicrobial should
be discontinued. A second drug to which the isolate is susceptible should be
substituted if the patient has not responded sufficiently; substitution is not
necessary if the patient response has been sufficient (Strohmeier et al.,
2014).
oIf resistance is reported to both antimicrobials and clinical evidence
of improvement is not evident, antimicrobial treatment should be changed to a
drug to which the offending organism is susceptible in vitro.
oIf resistance to the drug(s) that are used is reported but there has
been good clinical response, continuation with the initial therapy could be
considered, provided there are not other reasons (such as fluid therapy) that
might explain clinical improvement. Otherwise, a change in antimicrobial is
indicated.
o
Consultation
with a specialist (veterinary clinical microbiologist and/or veterinary
pharmacologist/pharmacist) is indicated with multidrug-resistant organisms (Strohmeier
et al., 2014).
A diagnosis other than bacterial pyelonephritis should be considered if
there is no improvement in systemic signs, hematology, or serum biochemistry
(e.g., azotemia, acute phase proteins) within 72 h of antimicrobial therapy and
the results of culture and susceptibility indicate susceptibility to the
antimicrobial used and there is confidence in client compliance. At that time,
consideration should be given to a diagnosis of subclinical bacteriuria (with
discontinuation of antimicrobial therapy) or for the presence of uncontrolled
underlying factors (e.g., ureteroliths, neoplasia) that would need to be
addressed to resolve the underlying infection (CLSI, 2023).
Treatment for 4–6 weeks has previously been recommended for pets (Weese et
al., 2011). However, the recommended duration of therapy for acute
bacterial pyelonephritis in humans is 7–14 days (Morello et al., 2016;
Ren et al., 2017). There is no reason to suspect that a longer duration
would be necessary for dogs and cats. In the absence of veterinary-specific
data, the Working Group recommends 10–14 days of treatment.
Subclinical
bacteriuria
Treatment of subclinical bacteriuria with antimicrobials is rarely
indicated and is discouraged. In animals where it is unclear whether clinical
signs are attributable to cystitis, a short course (e.g., 3–5 days duration) of
antimicrobials as recommended for sporadic cystitis could be considered. If
there is no clinical response, antimicrobials should be discontinued, as an
infectious process is unlikely. Treatment of animals with pyuria or other
cytological abnormalities without lower urinary tract signs is not recommended
(Johnstone, 2020).
The isolation of a multidrug-resistant bacterial species should not
affect the decision whether to treat subclinical bacteriuria. Antimicrobial
resistance genes are not virulence factors, and resistant organisms are not
more likely to cause disease than their susceptible counterparts.
Treatment of subclinical bacteriuria caused by multidrug-resistant
pathogens for infection control purposes (e.g., to eliminate urine shedding of
a possible pathogen) is not recommended. It is reasonable to assume that the
bacterial strain in the bladder is also present in the gastrointestinal tract;
therefore, even if bacteria are eliminated from the bladder with
antimicrobials, it would likely have limited impact on the overall risk .
In rare circumstances, treatment of subclinical bacteriuria may be
considered if there is concern that there is a particularly high risk of
ascending or systemic infection or that the bladder may be a focus of
extra-urinary infection.
In cases that are unable to display clinical signs of cystitis (e.g.,
spinal cord injury), a clinical judgment must be made, ensuring that
consideration of the need and potential adverse impacts (e.g., adverse drug
effects, antimicrobial resistance) are balanced. The relevance of changes in
urine appearance and odor (e.g., gross discoloration, malodor) to differentiate
infection from subclinical bacteriuria is unclear. However, sometimes a short
course of treatment (e.g., 3–5 days duration) could be considered for the
quality of life issues if bacteriuria may be playing a role (Johnstone, 2020).
Table 4. Drugs
for the management of bacterial urinary tract infection in dogs and cats (Weese
et al., 2011).
|
Drug (WHO category)a |
Dose |
Comments |
|
Amikacin (CIA) |
Dogs: 15–30 mg/kg IV/IM/SC every 24 h Cats: 10–14 mg/kg IV/IM/SC every 24 h |
Not recommended for routine use but may be useful
for treatment of MDR organisms. Potentially nephrotoxic. |
|
Amoxicillin (CIA) |
11–15 mg/kg PO every 8–12 h |
Good first-line option for sporadic bacterial
cystitis. Excreted in urine predominantly in active. Klebsiella spp.
are resistant. Not recommended for pyelonephritis. |
|
Amoxicillin/clavulanic acid (CIA) |
12.5–25 mg/kg PO every 12 h Note: dose of total
product (amoxicillin + clavulanic acid) |
Reasonable empiric choice for cystitis. Not
recommended for pyelonephritis. |
|
Ampicillin (CIA) |
N/A |
Not recommended because of poor oral
bioavailability. |
|
Cefazolin (HIA) |
22 mg/kg IV ∼30 min prior to the procedure. |
Main use is for peri-procedure prophylaxis as a
single pre-procedure dose |
|
Cefpodoxime proxetil (HP-CIA) |
Dogs: 5–10 mg/kg every 24 h PO days. Cats: no dose established. |
More active than cephalexin
or cefadroxil against Enterobacteria except Enterococcus spp. |
|
Ceftiofur (HP-CIA) |
Dogs: 2 mg/kg every 12–24 h SC Cats: no dose established |
Approved for treatment of bacterial cystitis in dogs
in some regions. Enterococcus spp. are resistant. |
|
Cefuroxime (HIA) |
Peri-operative prophylaxis: 20–50 mg/kg slow IV |
2nd generation cephalosporin that can be
used perioperatively. Enterococcus are
resistant. |
|
Cephalexin, cefadroxil (HIA) |
12–25 mg/kg PO every 12 h |
Narrow-spectrum activity; not active against
Enterobacterials. Enterococcus spp. are
resistant. |
|
Chloramphenicol (HIA) |
Dogs: 40–50 mg/kg PO every 8 h Cats: 12.5–20 mg/kg (to a maximum of 50 mg/cat) PO
every 12 h |
Reserved for multidrug
resistant infections with few other options. Myelosuppression can occur,
particularly in cats and with long-term (e.g. >28 days) therapy. Not a
first line treatment for pyelonephritis. |
|
Ciprofloxacin (HP-CIA) |
25–30 mg/kg PO every 24 h |
Sometimes used because of
lower cost than fluoroquinolones. Variable oral bioavailability. |
|
Doxycycline (HIA) |
5 mg/kg PO every 12 h |
Not excreted in urine at high levels but can achieve
levels that are effective against some pathogens. Care should be taken with
cats to reduce the risk of esophageal ulceration. |
|
Enrofloxacin (HP-CIA) |
5–20 mg/kg every 24 h (dogs) 5 mg/kg PO every 24 h (cats) |
Excreted in urine predominantly in active form.
Reserve for MDR infections but initial/empirical choice for pyelonephritis in
dogs. Not recommended for Enterococcus spp. It is recommended in cats. |
|
Fosfomycin (CIA) |
40 mg/kg PO (with food) every 12 h |
Should be reserved for multidrug resistant
infections. Do not use in cats. Potential option for pyelonephritis. |
|
Imipenem-cilastatin (CIA) |
5 mg/kg IV/IM every 6–8 h |
Reserve for treatment of MDR infections,
particularly those caused by ESBL-producing Enterobacterials or P.
aeruginosa, while E. faecium is inherently resistant. |
|
Levofloxacin (HP-CIA) |
25 mg/kg PO every 24 h (dogs) |
Sometimes used as a lower cost fluoroquinolone. High
oral bioavailability in dogs. |
|
Marbofloxacin (HP-CIA) |
2.7–5.5 mg/kg PO every 24 h |
Excreted in urine predominantly in active form. Good
first line choice for pyelonephritis. Not recommended for Enterococcus
spp. |
|
Meropenem (CIA) |
Dogs: 8.5 mg/kg every 12 h (SC) or every 8 h (IV) Cats: 10 mg/kg every 12 h IV, SC, IM |
Reserve for treatment of MDR infections,
particularly those caused by ESBL-producing Enterobacterials or P.
aeruginosa, while E. faecium is inherently resistant. |
|
Nitrofurantoin (IA) |
4.4–5 mg/kg PO every 8 h |
Option for sporadic bacterial cystitis, particularly
when MDR pathogens are involved. Excreted in urine predominantly in active
form. |
|
Orbifloxacin (HP-CIA |
Tablets: 2.5–7.5 mg/kg PO
every 24 h Suspension (cats): 7.5 mg/kg every 24 h |
Reserve for documented MDR
but good first choice for pyelonephritis. Not recommended for Enterococcus
spp. |
|
Pradofloxacin (HP-CIA) |
Dogs: 3–5 mg/kg PO every 24 h. Cats: 3–5 mg/kg once daily (tablets) or 5–7.5 mg/kg
every 24 h (suspension) |
Bacterial cystitis in dogs and cats. Greater
activity than older fluoroquinolones. Theoretically a good 1st
line choice for pyelonephritis, especially in cats. |
|
Trimethoprim-sulfadiazine/Trimethoprim-sulfamethoxazole/Ormetoprim-sulfadimethoxine
(HIA) |
15–30 mg/kg PO every 12 h |
Appropriate initial or empirical option. Concerns
regarding idiosyncratic and immune-mediated adverse effects in some patients;
however, this is most relevant with long-term therapy. If prolonged (>7
days) therapy is anticipated. Avoid in dogs that may be sensitive to
potential adverse effects such as hepatopathy, hypersensitivity and skin
eruptions. Activity against Enterococcus spp. in urine is
controversial and should be avoided. |
HP-CIA:
highest priority critically important antimicrobial. CIA: Critically important
antimicrobial.
HIA:
highly important antimicrobial. IA: Important antimicrobial.
CLSI:
Clinical Laboratory Standards Institute. ESBL: Extended spectrum β-lactamase.
a Drug
category per World Health Organization guidelines.
Treatment of subclinical bacteriuria caused by plaque-forming (Corynebacterium
urealyticum) and urease-producing (e.g., Staphylococci) organisms
could be considered because of their associations with encrusting cystitis and
struvite urolith formation, respectively (Biegen et al., 2013, Raab et
al., 2015). Because of the potential difficulties in treating these
conditions, consideration of a single short course (3–5 days duration) of
treatment, as per ‘Sporadic bacterial cystitis,’ could be considered after
confirming that bladder wall plaque or uroliths are not present. However, it is
unknown whether this is a necessary or effective approach. Continued treatment
of subclinical bacteriuria with these strains is likely not warranted.
There is currently no evidence that screening bacterial isolates for
urovirulence factors should impact decision-making for subclinical bacteria, as
there are currently no data that indicate the isolation of a bacterial strain
that possesses urovirulence genes is of greater clinical relevance to an
individual patient or that treatment will reduce the risk of disease (Johnstone, 2020).
There is currently no evidence that the use of adjunctive treatments
(e.g., cranberry extracts or probiotics) for the prevention of cystitis or
subclinical bacteriuria is effective, but there is no contraindication to the
use of treatments and supplements that are known to be safe.
If an animal that was recently diagnosed with subclinical bacteriuria
subsequently develops signs consistent with cystitis or pyelonephritis,
treatment designed to target the organism isolated while clinical signs were
absent can be considered. However, the likelihood that a subsequent infection
is caused by a previous bacteriuria isolate is not known and probably decreases
with time from the last culture. Repeat culture is indicated to determine the
optimal treatment if bacteriuria with a multidrug-resistant bacterial species
was previously diagnosed. If pyelonephritis is suspected, treatment targeting
the previously identified resistant bacterial isolate should be considered,
with a plan to de-escalate once culture results are available if a susceptible
organism is identified (Johnstone, 2020).
6.3. Antifungal
Drugs
Fluconazole is recommended as initial treatment in most cases because of
the high margin of safety, sensitivity of most strains of Candida spp.,
and excretion of active drug into urine in high concentrations. On the other
side, 19 Candida spp. other than C. albicans are more likely to
be resistant to fluconazole, and antifungal sensitivity testing is recommended
to determine if a higher dose of fluconazole is appropriate or if another drug
should be used. Although amphotericin B is renally excreted and achieves high
concentrations in urine, it is not often used because it is parenterally
administered and nephrotoxic (Reagan et al., 2019). Other commonly used
antifungal drugs, including itraconazole and ketoconazole, are not renally
excreted in active form (Hizlisoy et al., 2025). Secondary fungal UTI
occurs because of shedding of organisms into urine in cases with systemic
infections. Organisms most commonly associated with urine shedding are Aspergillus
spp. in dogs (particularly German shepherd dogs) and Cryptococcus spp.
in cats. These cases should be treated with antifungal agents standardly
recommended for systemic infections (Grassi et al., 2024).
6.4. Non
Antimicrobial UTIs Therapeutic Approaches
Multidrug-resistant bacterial isolates have significantly increased in
recent years as a cause of bacterial UTIs. This increasing resistance to
antimicrobial drugs leaves fewer therapeutic options for effective treatment.
Urinary tract infections with multidrug-resistant (MDR) bacteria increase
morbidity, treatment failures, and therapeutic costs. The emergence of
antibiotic-resistant bacteria in the community is necessitating the need to
explore non-pharmacologic treatment options to reduce the spread and proliferation
of these species (Amphaiphan et al., 2021).
Ozone therapy
Ozone is an emerging therapy with both potent antibacterial properties
and the ability to modulate the immune system, reduce oxidative stress induced
by chronic infection, and upregulate the endogenous antioxidant system,
providing further protection from free radical injury.
A 14-year-old female dog, paraplegic, suffered from recurrent bacterial
cystitis caused by Proteus spp. The case has been treated for two months
with enrofloxacin, amoxicillin, and potassium clavulanate. Because of the
development of adverse effects such as pharmacodermia and corneal ulcers, the
dog was referred to the ozone therapy sector of the Santa Maria Veterinary
Hospital, Brazil. The ozone therapy consisted of bladder instillations of
ozonized saline solution (O3SS); (59 μg/mL), once a day for 3 consecutive
days. After antisepsis of the external genitalia with 1% chlorhexidine
gluconate, a bladder catheterization was performed. Next, within a 20-minute
interval, the bladder was washed with 1 L of O3SS at a concentration
of 59 μg/mL. The data extracted from the study proposed removal of the biofilm
that covered the bladder mucosa, marked reduction in the suspended echogenic
content (sediment), and small hyperechogenic, punctiform, and linear structures
suggestive of crystals or small clots. Furthermore, it seemed that O3SS
had changed the pattern of susceptibility to cephalothin and neomycin, which
could expand the therapeutic options (Engelmann et al., 2023).
Probiotic therapy
Several probiotic bacteria have been proposed as an alternative to
combat MDR UTI. Lactic acid bacteria in the genus Lactobacillus are some
of the most studied and used probiotics (Gupta et al., 2024).
L. reuteri KUB-AC5 (AC5), isolated from the chicken gut, has been investigated for
both its direct and indirect effects against uropathogenic E. coli
(UPEC) isolates in vitro using a spot-on lawn, agar-well diffusion, and
competitive growth assays. It is found that viable AC5 cells and cell-free
components of this probiotic significantly reduced the UPEC growth of all
strains tested. The data showed that AC5 can attach to the examined cell line
and decrease UPEC attachment in a dose-dependent manner. Pretreatment of
UPEC-infected murine macrophage RAW264.7 cells with viable AC5 (multiplicity of
infection, MOI = 1) for 24 hours enhanced macrophage-killing activity and
increased proinflammatory and anti-inflammatory gene expression. These findings
indicate the gut-derived AC5 probiotic could be a potential urogenital
probiotic against MDR UTI (Tantibhadrasapa et al., 2024).
Snell et al. (2022) assessed the in vitro effects of E. coli
strain Nissle 1917 as a probiotic on UPEC isolated from 40 cats with clinical
UTI and subclinical bacteriuria. The results revealed that 52% of isolates were
found to be resistant to antimicrobials, with 19% of these being multidrug
resistant (MDR). Nissle 1917 adversely affected the growth of 82.5% of all
isolates and 100% of MDR isolates in vitro. The median zone of inhibition was
3.33 mm (range, 1.67 to 10.67 mm). Thirteen isolates were affected via
competitive overgrowth and 20 via growth inhibition.
The importance of dietary modifications in medical protocols designed to
treat and prevent LUTS in pets was postulated. The main goals of dietary
modifications to prevent LUTS are 1) promoting large dilute volumes of urine,
2) decreasing the relative supersaturation of urine for specific stone types,
and 3) promoting healthy bacterial populations in the gastrointestinal and
urogenital tracts. The impact of dietary composition, including dietary
moisture, protein concentration and digestibility, mineral concentrations,
inclusion of acidifiers and alkalinizing agents, inclusion of vitamin B6,
eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and γ-linolenic acid,
fiber concentration and characteristics, and oxalate degrading probiotics
(Kerr, 2013).
Bacteriophage
A study evaluated the therapeutic efficacy of phage P2-71 against P.
mirabilis, “a particular urinary tract pathogen,” in vivo and in vitro
environments. The results demonstrated that in vitro, bacteriophage P2-71
achieved significant reductions in P. mirabilis concentrations, with log
reductions of 1.537 and 0.7009 CFU/mL in laboratory and urine environments,
respectively (p < 0.001). The phage also decreased biofilm formation by
34-49% and lysed 15-25% of mature biofilms at various multiplicities of infection
(MOIs) (p < 0.001). In vivo, phage treatment significantly lowered bacterial
concentrations in the urine on Days 1 and 3 (p < 0.0001), achieving a
maximum reduction of 4.602 log₁₀ CFU/mL; however, its effectiveness diminished
by Day 5 (p > 0.05). Concurrently, phage titers decreased over time.
Importantly, phage treatment notably reduced bacterial load in the bladder,
kidneys, and spleen (p < 0.001). Inflammatory markers such as IL-6, IL-1β,
and TNF-α were significantly lower in the treatment group, especially in the
bladder (p < 0.0001), indicating an effective reduction in inflammation. So
it was concluded that bacteriophage P2-71 is a promising alternative therapy
for UTIs caused by MDR Proteus mirabilis (Wu et al., 2024).
6.5. Ancillary
Therapies and Prevention
Methenamine
Methenamine salt is a urinary antiseptic that is converted to
bacteriostatic formaldehyde in an acidic environment (urine pH <5.5). There
is controversy in human medicine as to whether methenamine prevents UTI,
although there is some evidence that it may be effective for short-term
prophylaxis. It is unknown if the 2 salts described in the literature,
hippurate and mandelate, are equally effective; the mandelate salt is difficult
to find. There is limited veterinary literature on the use of methenamine in small
animals, although there is a theoretic benefit. Studies of safety, efficacy,
and appropriate dosing are lacking. Commonly recommended doses are 10 to 20
mg/kg orally every 12 hours (dog) and 250 mg per cat orally every 12 hours.
Gastrointestinal upset and dysuria are the most commonly reported adverse
events; methenamine is poorly tolerated by feline patients. Methenamine should
not be used in cases of renal failure. Concurrent use of a urinary acidifier,
such as dl-methionine, is usually required for maximal effect (Weese et al.,
2011).
Cranberry
Proanthocyanidin, the “active ingredient” in cranberry, alters the
genotypic or phenotypic expression of fimbriae, which subsequently inhibits E.
coli adherence to human bladder and vaginal epithelial cells. There are few
veterinary studies in healthy dogs and no feline studies. In addition, quality
and potency are variable among over-the-counter products; ideally, each
formulation would be tested in the species of interest. The consensus of the
Antimicrobial Guidelines Working Group of the International Society for
Companion Animal Infectious Diseases is that there is insufficient evidence to
support the use of cranberry extract to prevent recurrent UTIs in dogs and cats
(Weese et al., 2011).
Anti-biofilms
Some bacteria have the capacity for biofilm formation, which facilitates
colonization. A biofilm is composed of organisms adhered together by a
self-produced polysaccharide matrix. It has been suggested that the bacteria
within the biofilm become sessile; they are protected from the immune system,
are antimicrobial, and inherently are resistant to shear forces of removal.
Biofilms are also implemented in the development of catheter-associated UTIs.
Strategies to prevent catheter-associated biofilms include using (1)
materials that are less amendable to biofilm formation and (2) coatings or
surface modifications that decrease biofilm formation. For example, silicone
catheters are preferred over latex because scanning electron microscope imaging
reveals that latex surfaces are more irregular and promote microbial adherence
(Olin and Bartges 2015). In a veterinary prospective study (n = 26 dogs)
evaluating biofilm formation on indwelling urinary catheters, sustained-release
varnish of chlorhexidine-coated urinary catheters statistically decreased
biofilm formation (Segev et al., 2013). There are an array of other
catheter coatings and modifications to decrease bacterial adherence and biofilm
formation that have primarily been studied in a research setting, including
silver coating, nanoparticles, iontophoresis, antimicrobials, urease and other
enzyme inhibitors, liposomes, and bacteriophages. Other novel strategies
include quorum sensing inhibitors and vibroacoustic stimulation (Siddiq and
Darouiche, 2012).
Vaccination
Vaccination is the most logical way to try and control infection and
reduce the prevalence of clinical disease and shedding. Bivalent vaccines
containing the serogroups L. icterohaemorrhagiae and L. canicola
have been in widespread use for many years, and infection with L. canicola
is now uncommon based upon the MAT. Leptospirosis has been reported in dogs in
Europe vaccinated with bivalent vaccines. Quadrivalent leptospirosis vaccines,
targeting L. canicola, L. icterohaemorrhagiae, L. pomona, and
L. grippotyphosa, were introduced in the USA in 2001 and are now
available in Europe. The current European vaccines contain either three
serogroups (L. canicola, L. icterohaemorrhagiae, and L.
grippotyphosa) or four serogroups (L. canicola, L.
icterohaemorrhagiae, L. grippotyphosa, and L. bratislava).
The European consensus statement recommends the use of quadrivalent vaccines
(Murphy, 2018).
Vaccination is recommended as soon as clinical recovery is seen. There
have been concerns raised about reactions to quadrivalent vaccines and about
reactions to leptospirosis vaccination, particularly in small dogs.
Veterinarians should report any suspected adverse drug reactions to the
authorities to increase the evidence available to confirm or refute these
claims. Current literature does not show vaccines with leptospiral antigen to
be associated with more significant reactions than other vaccines (Murphy,
2018). There is no commercial vaccine available for cats. However, one study
has shown that cats can produce antibodies (of lower titre magnitude than
vaccinated dogs) when experimentally inoculated with a commercial dog vaccine
(containing four different serovars). The follow-up time for the animals was 42
days, at which point only one animal maintained antibody levels. The authors of
that study suggest further work is needed before a vaccine against Leptospira
species for cats can be considered. Given the current lack of a vaccine, the
best way to avoid infection in cats is via prevention of exposure. Cats that
are kept indoors have a lower risk of being infected. Prevention of predation
opportunities and avoidance of contact with stagnant water, urine from infected
animals, and dogs at risk of clinical leptospirosis is recommended. For cats
that share an environment with a positively diagnosed animal, doxycycline can
be given at 5 mg/kg PO q12h or at 10 mg/kg PO q24h for 2 weeks. 19,60 (Murillo
et al., 2020).
TRANSMISSION
OF UROPATHOGENS BETWEEN HUMANS AND PETS: A PUBLIC HEALTH IMPORTANCE
Since the close relationship
and direct contact between humans and pets, the potential for transmission of
antimicrobial resistant bacteria or their resistance determinants from
companion animals to vulnerable populations, especially children and immunocompromised
persons, poses a public health concern (Amphaiphan et al., 2021). Given
the possibility of zoonotic transmission of antimicrobial-resistant bacteria,
veterinarians, when treating UTI cases, should inform pet owners of the
potential transmission risk (Yudhanto et al., 2022). Often, major
urinary bacterial pathogens of dogs can be resistant to antimicrobials commonly
used to treat UTIs or to antimicrobials important for human medicine. Previous
studies described resistance to carbapenems in E. coli isolates that
have carried the carbapenem resistance gene blaNDM-5. Carbapenems are
critically important antibiotics used to treat serious bacterial infections in
humans and are considered one of the human use last resorts (Tyson et al.,
2019). A recent study in Egypt declared the prevalence of shared UPEC serotypes
isolated from companion animals and humans, as well as sharing in the presence
of virulence determinants and the blaNDM-1 gene, which is responsible for
carbapenem resistance, proposing a potential public health concern (Hakim et
al., 2024a). In Australia, there is a particular significance with the
detection of the human UTI pandemic MDR E. coli strain O25b:H4-ST131 in
dogs. The significantly higher prevalence of this clonal lineage among fluoroquinolone-resistant
E. coli isolates from humans compared to dogs suggests that human-to-dog
transmission may currently predominate (Johnstone, 2020).
Similarly, resistance to
fluoroquinolones in Pseudomonas aeruginosa isolates has been mentioned
(Harada et al., 2012) and against cephalosporins (Hakim et al.,
2024b). Also, an increase in MDR methicillin-resistant Staphylococcus
pseudintermedius (MRSP) isolated from dogs with UTI has been reported
(Grönthal et al., 2017; Smith et al., 2020b).
The zoonosis, leptospirosis,
is caused by pathogenic spirochetes of the genus Leptospira, which
colonize the renal tubules where they reproduce before being excreted via the
urine. Infected urine or contaminated water are sources of leptospirosis
infection; asymptomatic and chronic carrier dogs can be maintenance hosts,
acting as sources of infection and therefore causing a public health problem.
Formerly, it was thought that domestic cats were resistant to leptospirosis
infection; however, recently published reports on feline leptospirosis conclude
that cats may play a role in the epidemiology of this disease (Donato et al.,
2022).
CONCLUSION
The illnesses of the
urinary tract in small animal, particularly in dogs and cats, occur frequently
with different etiologies. Many factors can predispose pets to UTIs, including
bladder stones, diabetes mellitus, anatomical abnormalities, and weakened immune
systems. The most common causes of UTIs are bacteria that go upwards through
the urethral opening. Bacterial UTIs are reported to occur in about 14% of all
dogs at some time during their lives, and the infection rate was higher in
females compared to males because of differences in anatomy. The infection rate
was highest in dogs younger than 2 years and older than 6 years, reaching 50%
in females older than 10 years. Due to their cleanliness and high urine
osmolarity, cats have a lower prevalence. The UTIs in dogs and cats are caused
by E. coli, P. aeruginosa, Klebsiella spp., Proteus
spp., Staphylococcus spp., Streptococcus spp., and Enterococci.
In addition to bacteria, fungi or viruses may also infect the urinary tracts.
Cystocentesis is the proper aseptic urine sampling for culture diagnostics.
Diagnosis is based on microbial culture as a gold standard method, besides
direct microscopy examination of urine sediment, biochemical examinations,
urinalysis, serological, and molecular techniques used for species
identification. The foundation of effective treatment is the disc-diffusion
method, which determines a pathogen's susceptibility to antibiotics; however,
the serial dilution technique is more precise. Use of empirical antibiotics is
substantial as they affect the most likely bacteria causing infection. In most
cases, amoxicillin, amoxicillin-clavulanic acid, and trimethoprim-sulfonamides
are considered the first empirical antimicrobial choices for UTI treatment in
pets. The increasing resistance to antimicrobial drugs leaves scarce
therapeutic choices for effective treatment. The emergence of MDR elevates the
need to explore non-pharmacologic treatment options such as ozone therapy,
probiotics, bacteriophage, herbs, and nutrient supplements. The increased
incidence of MDR bacteria represents a potential cause of antibiotic failure in
companion animals and constitutes a serious threat to global public health due
to the presence of potentially zoonotic microbial reservoirs in pet animals.
RECOMMENDATION
Urinary tract infections are a common issue in both dogs and cats, and
it's essential to understand how they're approached. Here's a breakdown of key
recommendations:
*Veterinary Consultation: If you suspect your pet has a UTI, seek
veterinary care immediately.
*Accurate Diagnosis: Ensure your veterinarian performs a urinalysis and,
ideally, a urine culture and sensitivity test.
*Follow Veterinary Instructions: Adhere strictly to your veterinarian's
treatment plan, including completing the full course of antibiotics.
*Preventative Measures: Provide plenty of fresh water and maintain a
clean litter box for cats.
*Advise vet. clinicians and vet. Lab specialists about the importance of
conducting bacterial culture and AST before starting UTI treatment to prevent
the emergence of MDR bacteria.
*Since pets could become the reservoirs of MDR bacteria that may be
transmitted to humans, veterinarians should inform pets’ owners about the
potential zoonotic transmission risk of these pathogens.
*Continuous monitoring of the AMR patterns of clinically important
bacterial urinary pathogens is warranted to identify emerging MDR strains.
*Monitoring the antibiotic resistance profiles in pet infections is
important not only for the public health implications but also to collect data
useful for the treatment of diseases in pets.
*To obtain information useful for global plans aimed at fighting AMR,
surveillance and rapid identification of AMR infections in pets have been
highlighted within the framework of the One Health approach, which recognizes
that the health of people is closely connected to the health of animals and the
shared environment.
REFERENCES
Acierno, M. (2024).
Making sense of the new UTI antimicrobial recommendations. Presented at Fetch
Coastal: Atlantic City, NJ. October 13, 2024. 395.
Álvarez-Pérez, M.E.,
García, M.T.C., María, L.F., María Á.D. and Teresa Peláez, J.L. (2016).
Acquired multi-azole resistance in Candida tropicalis during persistent
urinary tract infection in a dog. Sergio Blanco Medical Mycology Case Reports, 11, 9-12.
Alves, A.B.P., Dos Santos Machado, L., de
Souza, J.B., Spenchutt Vieira, G.P., Dias, T.S., de Almeida, V.L., do
Nascimento, E.R., Barreto, M.L., and Da Cunha, N. C. (2023). Detection of Mycoplasma
species at various anatomical sites of dogs from different types of kennels. Brazilian
Journal of Microbiology , 54(2) 1251–1255.
doi.org/10.1007/s42770-023-00947-x
Amphaiphan, C., Yano,
T., Som-in, M., Kungwong, P., Wongsawan, K. and Pusoonthornthum, R. (2021).
Antimicrobial drug resistance profile of isolated bacteria in dogs and cats
with urologic problems at Chiang Mai University Veterinary Teaching Hospital,
Thailand (2012–2016). Zoonoses Public Health, 68, 452–63.
Arnold, J.J., Hehn,
L.E. and Klein, D.A. (2016). Common questions about recurrent urinary tract
infections in women. Am. Fam. Physician, 93, 560–569.
Aso, S., Kitao, K.,
Hashimoto-Gotoh, A., Sakaguchi, S. and Miyazawa, T. (2021). Identification of
feline foamy virus-derived microRNAs. Microbes and Environments, 36(4),
ME21055. doi.org/10.1264/jsme2.ME21055
Bader, M.S., Loeb,
M., Leto, D., and Brooks, A.A. (2020). Treatment of urinary tract infections in
the era of antimicrobial resistance and new antimicrobial agents. Postgraduate
Medicine, 132(3), 234–250.
Baigi, S.R., Vaden,
S.L. and Olby, N.J. (2017). The frequency and clinical implications of
bacteriuria in chronically paralyzed dogs. J. Vet. Intern. Med., 31,
1790–1795.
Ballash, G.A., Mollenkopf, D.F., Diaz-Campos,
D., van Balen, J.C., Cianciolo, R.E. and Wittum, T.E. (2022). Pathogenomics and
clinical recurrence influence biofilm capacity of Escherichia coli
isolated from canine urinary tract infections. PloS one, 17(8),
e027046.
Balogh, O., Roch, M.,
Keller, S., Michel, E. and Reichler, I. (2017). The use of semi-quantitative
tests at Cesarean section delivery for the differentiation of canine fetal
fluids from maternal urine on the basis of biochemical characteristics. Theriogenology,
88,174–182. doi: 10.1016/j.theriogenology.2016.09.024.
Barot, H.M. Patel and
M.D. Parikh,P.V. (2022). Ultrasonographic diagnosis of urinary system
affections in dogs, Indian J. Vet. Sci. Biotech, 18 (4), 124–129.
Bartges, J. W. (2004). Diagnosis of urinary tract infections
Review article Veterinary Clinics of North America. Small Animal Practice 34,
(4), 923-933. https://doi.org/10.1016/j.cvsm.2004.03.001
Bennetta, P.F.,
Jessica, J.T., Martinb, P. Sarah, E.K., Mariano, M. and Vanessa, R. B. (2018). Long term
survival of a dog with disseminated Aspergillus deflectus infection
without definitive treatment. Medical
Mycology Case Reports, 22, 1-3.
Biasibetti, E., Maria
Teresa, C. M.T., Cocca, T., Bigliati, M., Bruni, N. and Martello, E. (2019). A
pilot study to evaluate alternative approaches for treatment of urinary tract
infections in dogs.
Asian Journal of Biomedical and Pharmaceutical
Sciences, 9 (66), 7-9.
Biegen, V.R.,
Slusser, P.G., Fischetti, A.J. and Geist, M.R. (2013). Successful treatment of
encrusted cystitis associated with Staphylococcus pseudintermedius
infection in the urinary bladder of a dog. J. Am. Vet. Med. Assoc., 242,
798–802.
Billips, B.K., Ryan,
E.,Y., John P.C., Anthony, J.S. and David, J. K. (2009). A Live-Attenuated
Vaccine for the Treatment of Urinary Tract Infection by Uropathogenic Escherichia
coli. The Journal of Infectious Diseases, 200 (2), 263–272, https://doi.org/10.1086/599839
Bleidorn, J.,
Hummers-Pradier, E., Schmiemann, G., Wiese, B. and Gágyor, I. (2016). Recurrent urinary tract
infections and complications after symptomatic versus antibiotic treatment:
follow-up of a randomised controlled trial. German Med. Sci., 14, Doc01.
Centers for Disease
Control and Prevention. Urinary Tract Infection (Catheter Associated Urinary
Tract Infection [CAUTI] and Non-Catheter Associated Urinary Tract Infection
[UTI] and Other Urinary System Infection (USI) Events). Available online:
Chou, H. I., Chen,
K.S., Wang, H.C. and Lee, W.M. (2016). Effects of cranberry extract on
prevention of urinary tract infection in dogs and on adhesion of Escherichia
coli to Madin-Darby canine kidney cells. Am. J. Vet. Res., 77,
421–427.
Clinical Laboratory
Standards Institution (2023): Performance standards for Antimicrobial Disc and Dilution
Susceptibility Testing; Approved Standard-33rd Edition. CLSI
supplement M100-A3. Wayne Pennsylvania, USA.
Curtis, K.M., Foster,
P.C., Smith, P.S., Monn, M.P., Stillman, B.A., Chandrashekar, R., Lappin, M.R.
and Goldstein, R.E. (2015). Performance of a Recombinant LipL32 Based Rapid
In-clinic ELISA (SNAP® Lepto) for the detection of antibodies against
Leptospira in Dogs. Intern. J.
Appl. Res. Vet. Med., 13 (3), 182-189.
D’Anjou, M. A.,
Bédard, A. and Dunn, M.E. (2011). Clinical significance of renal pelvic
dilatation on ultrasound in dogs and cats. Vet. Radiol. Ultrasound., 52,
88–94.
Dahlem, D.P., Neiger,
R., Schweighauser, A., Francey, T., Yerramilli, M., Obare, E. and Steinbach,
S.M.L. (2017). Plasma symmetric dimethylarginine concentration in dogs with
acute kidney injury and chronic kidney disease. J. Vet. Intern. Med., 31, 799-804.
Delaude, A.,
Rodriguez-Campos, S., Dreyfus, A., Counotte, M.J., Francey, T., Schweighauser,
A., Lettry, S. and Schuller, S. (2017). Canine leptospirosis in Switzerland-A
prospective cross-sectional study examining seroprevalence, risk factors and
urinary shedding of pathogenic leptospires. Preventive Veterinary Medicine,
141, 48–60.
Deprey, J., Baldinger, A., Livet, V.,
Blondel, M., Taroni, M., Lefebvre, C., Goy-Thollot, I., Moissonnier, P.,
Viguier, É., Pouzot-Nevoret, C., Carozzo, C. and Cachon, T. (2021). Risk
factors and clinical relevance of positive urine cultures in cats with
subcutaneous ureteral bypass. BMC Veterinary Research, 17(1), 199. https://doi.org/10.1186/s12917-021-02898-7
Donato, G., Masucci,
M., Hartmann, K., Goris, M.G. A., Ahmed, A. A., Archer, J., Alibrandi, A. and
Pennisi, M.G. (2022). Leptospira spp. Prevalence in cats from Southern
Italy with evaluation of risk factors for exposure and clinical findings in
infected cats. Pathogens, 11(10), 1129.
Dorsch, R., Salgado,
M. and Monti, G., (2017). Urine shedding of pathogenic Leptospira spp.
in cats. Proceedings of the 10th International Leptospirosis Society
Conference; 21-24; Palmerston North, New Zealand.
Eissa, N. (2023).
Fungal diseases of dogs and cats In book: Introduction to Diseases, Diagnosis,
and Management of Dogs and Cats Publisher: Elsevier doi /
10.1016/B978-0-443-18548-9.00035-4.
Engelmann, A. M.,
Bueno, A., Barbosa, N.V., Machado, C., Correa, D., Cargnelutti, J.F. and de
Andrade, C.M. (2023). Effectiveness of ozonized saline solution in the
treatment of Proteus spp. bacterial cystitis. Medical Gas Research,
13(3), 155–158. https://doi.org/10.4103/2045-9912.350861
Englar R. E. (2022).
(Canine and Feline Practice), Sharon M. Dial DVM, PhD, DACVP (Clinical and
Anatomic Pathology) In book:
Low‐Cost Veterinary Clinical Diagnostics. doi: 10.1002/9781119714521.ch12.
Esparaz, A.M., Pearl,
J.A., Herts, B.R., LeBlanc, J. and Kapoor, B. (2015). Iatrogenic urinary tract
injuries: etiology, diagnosis, and management. Semin. Intervent. Radiol., 32(2),195-208.
doi: 10.1055/s-0035-1549378.
Facchin, A., Filipe, J., Mauri, I.,
Tagliasacchi, F., Grilli, G., Vitiello, T., Ratti, G., Musa, L., Penati, M.,
Scarpa, P. and Lauzi, S. (2025). Antimicrobial Resistance and Biofilm-Forming
Ability in ESBL-Producing and Non-ESBL-Producing Escherichia coli and Klebsiella
pneumoniae Isolated from Canine Urinary Samples from Italy. Antibiotics,
14(1), 31.
Farag, H.S., Ali,
M.E., Abdel Masseih, E.S. and Bakry, N.M. (2024). Diagnostic ultrasonography
and antimicrobial resistance of different pathogens associated with canine and
feline lower urinary tract disorders. Comparative Immunology, Microbiology and Infectious Diseases, 112, 2024: 102216.
Gágyor, I., Bleidorn,
J., Kochen, M.M., Schmiemann, G., Wegscheider, K. and HummersPradier, E.
(2015). Ibuprofen versus fosfomycin for uncomplicated urinary tractinfection in
women: randomised controlled trial. BMJ, 351, h6544.
Garcês, A., Lopes,
R., Silva, A., Sampaio, F., Duque, D. and Brilhante-Simões, P. (2022).
Bacterial isolates from urinary tract infection in dogs and cats in Portugal,
and their antibiotic susceptibility pattern: a retrospective study of 5 years
(2017–2021). Antibiotics, 11(11), 1520.
Govindarajan, D.K., Eskeziyaw, B.M.,
Kandaswamy, K. and Mengistu, D.Y. (2024). Diagnosis of extraintestinal
pathogenic Escherichia coli pathogenesis in urinary tract infection. Current
Research in Microbial Sciences, 7, 100296. https://doi.org/10.1016/j.crmicr.2024.100296
Grant, D.C.,
Keebaugh, A.E. and Werre, S. (2023). Point-of-care rapid immunoassay performed
on voided urine, refrigerated up to 24 hours, accurately detects bacteriuria. Journal
of the American Veterinary Medical Association, 262(1), 88–92.
https://doi.org/10.2460/javma.23.07.0408
Grant, D.C., Nappier, M.T. and Corrigan, V.K.
(2021). Diagnostic accuracy of a point-of-care test using voided urine samples
for detection of bacteriuria in dogs with signs of lower urinary tract disease.
J. Vet. Intern. Med., 35(2),993-996. doi: 10.1111/jvim.16040.
Grassi, A.,
Cafarchia, C., Decaro, N., Rhimi, W., De Laurentiis, V., D'Annunzio, G., Luppi,
A. and Prati, P. (2024). Systemic Candida Infection and Pulmonary
Aspergillosis in an Alpaca (Vicugna pacos): A Case Report. Journal of
Fungi (Basel, Switzerland), 10(3), 227. https://doi.org/10.3390/jof10030227
Grönthal. T., Eklund,
M., Thomson, K., Piiparinen, H., Sironen, T. and Rantala, M. (2017).
Antimicrobial resistance in Staphylococcus pseudintermedius and the
molecular epidemiology of methicillin-resistant S. pseudintermedius in
small animals in Finland. J. Antimicrob. Chemother., 72, 1021–30.
10.1093/jac/dkx086
Gupta, V.,
Mastromarino, P. and Garg, R. (2024). Effectiveness of prophylactic oral and/or
vaginal probiotic supplementation in the prevention of recurrent urinary tract
infections: A randomized, double-blind, placebo-controlled trial. Clinical
infectious diseases : an official publication of the Infectious Diseases
Society of America, 78(5), 1154–1161.
https://doi.org/10.1093/cid/ciad766
Hakim, A., Khalaf,
D.D., Farahat, E., Mohammed, M.D., El-Dabae, W.H., El-Razik, K.A., Dapgh, A.N.,
Fouad, E.A. and Abuelhag, H. (2024a). Emergence of carbapenem resistant Escherichia
coli isolated from human and companion animals associated with urinary
tract infection. Adv. Anim. Vet. Sci., 12(s1), 127-138.
doi.org/10.17582/journal.aavs/2024/12.s1.127.138
Hakim, A.S., Dorgham,
S.M., Abuelhag, H.A., Sadek, E.G., Dapgh, A.N., Youssif, Nesma H. and Fouad, E.
A. (2024b). Isolation and identification of Pseudomonas aeruginosa
obtained from dogs and cats in Great Cairo regarding status of phenotypic
antimicrobial resistance pattern. Egyptian Pharmaceutical Journal, 23(3),
525-531, 10.4103/epj.epj_340_23
Halaji, M., Fayyazi,
A., Rajabnia, M., Zare, D., Pournajaf, A. and Ranjbar, R. (2022). Phylogenetic
group distribution of uropathogenic Escherichia coli and related
antimicrobial resistance pattern: A meta-analysis and systematic review. Frontiers
in Cellular and Infection Microbiology,12, 790184.
Harada, K., Arima,
S., Niina, A., Kataoka, Y. and Takahashi, T. (2012). Characterization of Pseudomonas
aeruginosa isolates from dogs and cats in Japan: current status of
antimicrobial resistance and prevailing resistance mechanisms. Microbiol
Immunol., 56,123–127. doi:10.1111/j.1348-0421.2011.00416.x
Hedström, M., Møller,
M., Patsekhina, H., Damborg, P., Jessen, L. R. and Sørensen, T. M. (2021). The
effect of urine storage temperature and boric acid preservation on quantitative
bacterial culture for diagnosing canine urinary tract infection. BMC
Veterinary Research, 17(1), 379.
Hemmatzadeh, F.,
Niap, F., Bennett, B. A., Trott, D. J. and Peaston, A.E. (2019). A novel
quantitative polymerase chain reaction to monitor urinary tract mycoplasma
infection in a dog. Letters in Applied Microbiology, 68(5),
409–414. https://doi.org/10.1111/lam.13139.
Herbert, J., Chong,
D., Spielman, D., Krockenberger, M., Wildner, J., and Bishop, R. (2019).
Unusual presentation and urinary tract obstruction due to disseminated
intra-abdominal eumycetomas caused by Curvularia species in a
dog. Medical Mycology Case Reports, 26, 28–31.
https://doi.org/10.1016/j.mmcr.2019.09.002
Hernando, E., Vila, A., D'Ippolito, P., Rico,
A. J., Rodon, J. and Roura, X. (2021). Prevalence and characterization of
urinary tract infection in owned dogs and cats from Spain. Topics in
Companion Animal Medicine, 43, 100512. https://doi.org/10.1016/j.tcam.2021.100512
Hizlisoy, H., Dishan,
A., Bekdik, I.K., Barel, M., Koskeroglu, K., Ozkaya, Y., Aslan, O. and Yilmaz,
O.T. (2025). Candida albicans in the oral cavities of pets: biofilm
formation, putative virulence, antifungal resistance profiles and
classification of the isolates. International microbiology: The Official
Journal of the Spanish Society for Microbiology, 28(3), 423–435. https://doi.org/10.1007/s10123-024-00552-4
Holzapfel, M.,
Taraveau, F. and Djelouadji, Z. (2021). Serological and molecular detection of
pathogenic Leptospira in domestic and stray cats on Reunion Island,
French Indies. Epidemiology and Infection, 149, e229.
doi.org/10.1017/S095026882100176X.
Jessen, L.R.,
Sørensen, T.M., Bjornvad, C.R., Nielsen, S.S. and Guardabassi, L. (2015).
Effect of antibiotic treatmentin canine and feline urinary tractinfections: a
systematic review. Vet. J., 203, 270–277.
Johnstone, T. (2020).
A clinical approach to multidrug-resistant urinary tract infection and
subclinical bacteriuria in dogs and cats. N. Z. Vet. J., 68, 69–83. doi:
10.1080/00480169.2019.1689196.
Karah, N., Rafei, R.,
Elamin, W., Ghazy, A., Abbara, A., Hamze, M. and Uhlin, B. E. (2020). Guideline
for urine culture and biochemical identification of bacterial urinary pathogens
in low-resource settings. Diagnostics, 10(10), 832.
doi.org/10.3390/diagnostics10100832
Kerr, K.R. (2013).
Companion Animals Symposium: dietary management of feline lower urinary tract
symptoms. Journal of Animal Science, 91(6), 2965–2975.
https://doi.org/10.2527/jas.2012-6035.
Kocúreková, T., Koščová, J. and Hajdučková,
V. (2021). Infections of the urinary tract of bacterial
origin in dogs and cats. Folia Veterinaria, 65 (1), 59-66
doi.org/10.2478/fv-2021-0008.
Kruger, J.M., Osborne, C.A. and Wise, A.G. (2011). Viruses and urinary tract disease. In: Polzin
D, Bartges JW, editors. Nephrology and urology of small animals. Chichester
(United Kingdom): Blackwell Publishing Ltd; 725–33.
L'Abée-Lund, T.M., Heiene, R., Friis, N.F.,
Ahrens, P. and Sørum, H. (2015). Mycoplasma
canis and urogenital disease in dogs in Norway. Vet. Rec., 153,
231–235.
Lee, B. S., Bhuta,
T., Simpson, J.M. and Craig, J.C.
(2012). Methenamine hippurate for preventing urinary tract infections. The
Cochrane Database of Systematic Reviews, 10(10), CD003265.
Lien, C.J. and Wang,
S.L. (2023). The impact of urine storage methods on the results of quantitative
bacterial culture in dog and cat. Veterinarni Medicina, 68(3),
116–121. doi.org/10.17221/111/2022-VETMED
Lippi, I.,
Habermaass, V., Gori, E., Ebani, V.V., Pierini, A., and Marchetti, V. (2022).
Urinary cytology: Potential role in canine urinary tract infections. Veterinary
Sciences, 9(6), 304. doi.org/10.3390/vetsci9060304
Lusby, A.L., Kirk,
C.A., JW, Moyers, T.D., Toll, P.W. (2011). Prevalence of asymptomatic bacterial
urinary tract infections in morbidly obese dogs. Proceedings of the American
College of Veterinary Internal Medicine Forum, Denver, CO, 15th–18th
June 2011.
Mansour, A., Hariri,
E., Shelh, S., Irani, R. and Mroueh, M. (2014). Efficient and cost effective
alternative treatment for recurrent urinary tract infections and interstitial
cystitis in women: a two-case report. Case Rep. Med., 2014, 698758.
Mantis, P. (2008). Ultrasonography of the urinary and
genital system of the dog and cat, Iran. J. Vet. Surg. Suppl., 2008,
63–71.
McGhie, J.A., Stayt,
J. and Hosgood, G.L. (2014). Prevalence of bacteriuria in dogs without clinical
signs of urinary tract infection presenting for elective surgical procedures. Aust.
Vet. J., 92, 33–37.
McGuire, N.C.,
Schulman, R., Ridgway, M.D. and Bollero, G. (2002). Detection of occult urinary
tract infections in dogs with diabetes mellitus. J. Am. Med. Assoc., 38, 541-544.
McMurdo, M.E., Argo,
I., Phillips, G., Daly, F. and Davey, P. (2009). Cranberry or trimethoprim for
the prevention of recurrent urinary tract infections? A randomized controlled
trial in older women. The Journal of Antimicrobial Chemotherapy, 63(2),
389–395. https://doi.org/10.1093/jac/dkn489
Melandri M., Veronesi
M.C. and Alonge S. (2020). Urinalysis in Great Dane puppies from birth to 28
days of age. Animals (Basel). 7;10(4),636. doi: 10.3390/ani10040636.
Melgarejo, T.,
Oakley, B. B., Krumbeck, J. A., Tang, S., Krantz, A. and Linde, A. (2021).
Assessment of bacterial and fungal populations in urine from clinically healthy
dogs using next-generation sequencing. Journal of Veterinary Internal
Medicine, 35(3), 1416–1426. doi.org/10.1111/jvim.16104
Miotto, B.A.,
Guilloux, A.G. A., Tozzi, B.F., Moreno, L.Z., da Hora, A.S., Dias, R.A.,
Heinemann, M.B., Moreno, A.M., Filho, A.F.S., Lilenbaum, W. and Hagiwara, M.K.
(2018). Prospective study of canine leptospirosis in shelter and stray dog
populations: Identification of chronic carriers and different Leptospira species
infecting dogs. PloS one, 13(7), doi.org/10.1371/journal.pone.0200384.
Morello, W., La
Scola, C., Alberici, I. and Montini, G. (2016). Acute pyelonephritis in
children. Pediatric Nephrology (Berlin, Germany) vol. 31(8), 1253-65.
doi:10.1007/s00467-015-3168-5
Murillo, A., Goris,
M., Ahmed, A., Cuenca, R. and Pastor, J. (2020). Leptospirosis in cats: Current
literature review to guide diagnosis and management. J. Feline Med. Surg.,
22(3),216-228. doi: 10.1177/1098612X20903601.
Murphy, K. (2018).
Leptospirosis in dogs and cats: new challenges from an old bacteria. In
Practice, 40 (6), 218-229. https://doi.org/10.1136/inp.k2926
Nicolle, L.E.,
Bradley, S., Colgan, R., Rice, J.C., Schaeffer, A. and Hooton, T.M. (2005). Infectious diseases society of
America guidelines for the diagnosis and treatment of asymptomatic bacteriuria
in adults. Clin. Infect. Dis., 40, 643–654.
O’Neil, E., Horney,
B., Burton, S., Lewis, P.J., MacKenzie, A. and Stryhn, H. (2013). Comparison of wet-mount,
Wright-Giemsa and Gram-stained urine sediment for predicting bacteriuria in
dogs and cats. Can. Vet. J., 54, 1061–1066.
Olby, N.J., Vaden,
S.L., Williams, K., Griffith, E.H., Harris, T., Mariani, C.L., Muñana, K. R., Early, P.J., Platt, S.R. and
Boozer, L. (2017). Effect of cranberry extract on the frequency of bacteriuria
in dogs with acute thoracolumbar disk herniation: a randomized controlled
clinical trial. J. Vet. Intern. Med, 31, 60–68.
Olin, S.J. and
Bartges, J.W. (2015). Urinary tract infections: treatment/comparative
therapeutics. Vet. Clin. North Am. Small Anim. Pract., 45(4), 721-746.
doi: 10.1016/j.cvsm.2015.02.005.
Palerme, J.,
Lamperelli,E. and Gagne, J. (2019). Seroprevalence of Leptospira spp., Toxoplasma
gondii, and Dirofilaria immitis in free-roaming cats in Iowa. Vector
Borne Zoonotic Dis., 19, 193–198.
Patterson, C.A.,
Bishop, M.A., Pack, J.D., Cook, A.K. and
Lawhon, S.D. (2016). Effects of processing delay, temperature, and
transport tube type on results of quantitative bacterial culture of canine
urine. J. Am. Vet. Med. Assoc., 248, 183–187.
Pereira, A., Baptista, C.J., Oliveira, P.A.
and Coelho, A. (2024).
Urinary tract bacterial infections in small animal practice: clinical and Epidemiological
Aspects. Veterinarska Stanica ,55 (6), 703-716.
doi.org/10.46419/vs.55.6.8.
Peterson, A.L.,
Torres, S.M.F., Rendahl, A. and Koch, S.N. (2012). Frequency of urinary tract
infection in dogs with inflammatory skin disorders treated with ciclosporin
alone or in combination with glucocorticoid therapy: a retrospective study. Vet. Dermatol., 23, 201, e243.
Public Health
England. UK Standards for Microbiology Investigations. Investigation of urine.
B 41 Issue 8.7. Available online:
https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/770688/B_41i8.7.pdf
(accessed on 21 August 2020).
Puchot, M.L., Cook,
A.K. and Pohlit, C. (2017). Subclinical bacteriuria in cats: prevalence, findings on contemporaneous
urinalyses and clinical risk factors. J. Feline Med. Surg,. 19, 1238–1244.
Punia, M., Kumar, A., Charaya, G. and Kumar,
T. (2018). Pathogens isolated from clinical cases of
urinary tract infection in dogs and their antibiogram. Vet World, 11(8),1037-1042.
doi: 10.14202/vetworld.2018.1037-1042.
Raab, O., Béraud, R.,
Tefft, K.M. and Muckle, C.A. (2015). Successful treatment of Corynebacterium
urealyticum encrusting cystitis with systemic and intravesical
antimicrobial therapy. Can. Vet. J., 56, 471–475.
Rampacci, E.,
Bottinelli, M. Stefanetti, V. Hyatt, D.R. Sgariglia, E. and Coletti, M. (2018).
Antimicrobial susceptibility survey on bacterial agents of canine and feline
urinary tract infections: weight of the empirical treatment, J. Glob.
Antimicrob. Resist., 13, 192–196.
Reagan, K.L., Dear,
J.D., Kass, P.H. and Sykes, J.E. (2019). Risk factors for Candida urinary tract
infections in dogs and cats. Journal of Veterinary Internal Medicine, 33(2),
648–653. doi.org/10.1111/jvim.15444
Reine, N.J. and
Langston, C.E. (2005). Urinalysis interpretation: how to squeeze out the
maximum information from a small sample. Clinical Techniques in Small Animal
Practice, 20(1), 2–10. doi.org/10.1053/j.ctsap.2004.12.002
Ren, H., Li, X., Ni,
Z.-H., Niu, J.-Y., Cao, B., Xu, J., Cheng, H., Tu, X.-W., Ren, A.-M. and Hu, Y.
(2017). Treatment of complicated urinary tract infection and acute
pyelonephritis by short-course intravenous levofloxacin (750 mg/day) or
conventional intravenous/oral levofloxacin (500 mg/day): prospective,
openlabel, randomized, controlled, multicenter, non-inferiority clinical trial.
Int. J. Urol. Nephrol., 49, 499–507.
Reppas, G. and
Foster, S.F. (2016). Practical urinalysis in the cat 1: Urine macroscopic
examination ‘tips and traps’. Journal of Feline Medicine and Surgery, 18,
190–202.
Rodrigues, F., Maia,
M.J., das Neves, J., Sarmento, B., Amaral, M.H. and Oliveriera, M.B.P.P.
(2014).Vaginal suppositories containing Lactobacillus acidophilus:
development and characterization. Drug Dev. Ind. Pharm., 41, 1518–1525.
Schuller, S.,
Francey, T. and Hartmann, K. (2015). European consensus statement on
leptospirosis in dogs and cats. J Small Anim. Pract., 56, 159–179.
Segev, G., Bankirer,
T. and Steinberg, D. (2013). Evaluation of urinary catheters coated with
sustained-release varnish of chlorhexidine in mitigating biofilm formation on
urinary catheters in dogs. J. Vet. Intern. Med., 27,39–46. doi:
10.1111/j.1939-1676.2012.01027.x
Segev, G., Sykes,
J.E., Klumpp, D.J., Schaeffer, A.J., Antaki, E.M., Byrne, B.A., Yaggie, R. E. and Westropp, J.L. (2018).
Evaluation of the live biotherapeutic product, asymptomatic bacteriuria Escherichia
coli 2-12, in healthy dogs and dogs with clinical recurrent UTI. J. Vet.
Intern. Med., 32, 267–273.
Sender, D., Hulsey, B., Cañete-Gibas, C.,
Wiederhold, N., Lee, J.K., Finley, A., Cruz, C. and White, M.E. (2024). Disseminated Aspergillus citrinoterreus and
concurrent localized dermal phaeohyphomycosis in an immunosuppressed dog. Clin.
Case Rep., 12(2),e7573. doi: 10.1002/ccr3.7573.
Shropshire, S.B.,
Veir, J.K. and Morris, A.K. (2016). Evaluation of the Leptospira species
microscopic agglutination test in experimentally vaccinated cats and Leptospira
species seropositiv-ity in aged azotemic client-owned cats. J. Feline Med.
Surg., 18, 768–772.
Siddiq, D.M. and
Darouiche, R.O. (2012). New strategies to prevent catheter-associated urinary
tract infections. Nat. Rev. Urol., 9, 305–314. doi:
10.1038/nrurol.2012.68.
Smee, N., Loyd, K.
and Grauer, G. F. (2013). UTIs in small animal patients: part 2: diagnosis,
treatment, and complications. Journal of the American Animal Hospital
Association, 49(2), 83–94. doi.org/10.5326/JAAHA-MS-5944.
Smith, J.M.,
Thomason, C., Sun, X. and Lennon, E.M. (2020a). Diagnosis of bacterial urinary
tract infection: Utility of urine myeloperoxidase concentration to predict
urine culture results in dogs. PloS one, 15(5), e0233566.
doi.org/10.1371/journal.pone.0233566
Smith, J.T., Amador,
S., McGonagle, C.J., Needle, D., Gibson, R. and Andam, C.P. (2020b). Population
genomics of Staphylococcus pseudintermedius in companion animals in the
United States. Commun Biol., 3, 282. 10.1038/s42003-020-1009-y
Smoglica, C.,
Evangelisti, G., Fani, C., Marsilio, F., Trotta, M., Messina, F. and Di
Francesco, C.E. (2022). Antimicrobial Resistance Profile of Bacterial Isolates
from Urinary Tract Infections in Companion Animals in Central Italy. Antibiotics
(Basel), 11(10),1363. doi: 10.3390/antibiotics11101363.
Snell, C.B., Winston,
J.A., Quimby, J.M., Diaz-Campos, D., Gibson, J.F., Harrison, A., Byron, J.M.,
Justice, S.S. and Rudinsky, A.J. (2022).
Escherichia coli probiotic exhibits in vitro growth-limiting effects on
clinical feline uropathogenic E coli isolates. American Journal of
Veterinary Research, 83(7), ajvr.21.07.0096.
doi.org/10.2460/ajvr.21.07.0096
Soliman, E.A., Saad,
A., Abd El Tawab, A.A., Elhofy, F. I., Rizk, A.M., Elkhayat, M., Kozytska, T.,
Ilyas, M., Bassiouny, M., Brangsch, H., Pletz, M.W., Neubauer, H., Sprague,
L.D. and Wareth, G. (2024). Exploring AMR and virulence in Klebsiella
pneumoniae isolated from humans and pet animals: A complement of phenotype
by WGS-derived profiles in a One Health study in Egypt. One Health, 19,
100904. doi.org/10.1016/j.onehlt.2024.100904
Sørensen, T.M.,
Jensen, A.B., Damborg, P., Bjørnvad, C.R., Guardabassi, L. and Jessen, L.R.
(2016). Evaluation of different sampling methods and criteria for diagnosing
canine urinary tract infection by quantitative bacterial culture. Veterinary
Journal (London, England : 1997), 216, 168–173.
doi.org/10.1016/j.tvjl.2016.08.007
Stapleton, A. E.,
Au-Yeung, M., Hooton, T. M., Fredricks, D. N., Roberts, P. L., Czaja, C. A.,
Yarova-Yarovaya, Y., Fiedler, T., Cox, M. and Stamm, W. E. (2011). Randomized,
placebo-controlled phase 2 trial of a Lactobacillus crispatus probiotic
given intravaginally for prevention of recurrent urinary tract infection.
Clinical infectious diseases: An official publication of the Infectious
Diseases Society of America, 52(10), 1212–1217. doi.org/10.1093/cid/cir183
Strachan, N. A.,
Hales, E. N. and Fischer, J. R. (2022). Prevalence of positive urine culture in
the presence of inactive urine sediment in 1049 urine samples from dogs. Journal
of Veterinary Internal Medicine, 36(2), 629–633. doi.org/10.1111/jvim.16378
Strohmeier, Y.,
Hodson, E.M., Willis, N.S., Webster, A.C. and Craig, J.C. (2014). Antibiotics
for acute pyelonephritis in children. Cochrane Database Syst. Rev. CD003772.
Sung, J.I.,
won-Kyoung, Y. and Changbaig, H. (2017). Candida albicans urinary tract
infection in a Shih Tzu dog with immune-mediated hemolytic anemia. Korean Journal of Veterinary
Research, 57(2),139-141 doi.org/10.14405/kjvr.2017.57.2.139
Sutter, C.M., Dear,
J.D., Fine, J.R., Pires, J., Sykes, J.E., Segev, G. and Westropp, J.L. (2023).
Evaluation of a rapid immunoassay for bacteriuria in dogs. Journal of
Veterinary Internal Medicine, 37(3), 1015–1020.
doi.org/10.1111/jvim.16684.
Swenson, C.L.,
Boisvert, A.M. and Kruger, J.M.
(2004). Evaluation of modified Wright-staining of urine sediment as a method
for accurate detection of bacteriuria in dogs. J. Am. Vet. Med. Assoc., 224
, 1282-1289.
Sykes, J.E. (2014). Candidiasis. Canine feline infect. dis. St.
Louis: Elsevier. 653–9. doi: 10.1016/B978-1-4377-0795-3.00067-3.
Sykes, J.E.,
Hartmann, K., Lunn, K.F., Moore, G.E., Stoddard, R.A. and Goldstein, R.E.
(2011). 2010 ACVIM small animal consensus statement on leptospirosis:
diagnosis, epidemiology, treatment, and prevention. J. Vet. Intern. Med., 25(1),1-13.
doi: 10.1111/j.1939-1676.2010.0654.x.
Tantibhadrasapa, A.,
Li, S., Buddhasiri, S., Sukjoi, C., Mongkolkarvin, P., Boonpan, P., Wongpalee,
S. P., Nakphaichit, M., Nitisinprasert, S., Hsieh, M. H. and Thiennimitr, P.
(2024). Probiotic Limosilactobacillus reuteri KUB-AC5 decreases
urothelial cell invasion and enhances macrophage killing of uropathogenic Escherichia
coli in vitro study. Frontiers in Cellular and Infection Microbiology,
14, 1401462.
Temmerman, R., Berlamont, H., El Garch, F.,
Rose, M., Simjee, S., Meschi, S. and de Jong, A. (2024). Antimicrobial
susceptibility of canine and feline urinary tract infection pathogens isolated
from animals with clinical signs in European veterinary practices during the
period 2013-2018. Antibiotics (Basel, Switzerland), 13(6), 500.
doi.org/10.3390/antibiotics13060500
Teng, L., Feng, M.,
Liao, S., Zheng, Z., Jia, C., Zhou, X., Nambiar, R. B., Ma, Z. and Yue, M.
(2023). A Cross-Sectional Study of Companion Animal-Derived Multidrug-Resistant
Escherichia coli in Hangzhou, China. Microbiology Spectrum, 11(2),
e0211322. https://doi.org/10.1128/spectrum.02113-22
Tigabie, M., Ayalew,
G. and Demoze, L. (2025). Extended-spectrum beta-lactamase and
carbapenemase-producing Gram-negative bacteria in urinary tract infections in
Ethiopia: a systematic review and meta-analysis. BMC Urol., 25, 11.
doi.org/10.1186/s12894-025-01695-w
Tsoi, M., Magenta,
F., Kline, A., Conkling A., Rinosh M. and Sarah, M. (2021) Scedosporium
apiospermum infection presenting as a mural urinary bladder mass and focal
peritonitis in a Border Collie Corner. Medical Mycology Case Reports, 33,
9–13.
Tyson, G.H., Li, C.,
Ceric, O., Reimschuessel, R., Cole, S. and Peak, L. (2019). Complete genome
sequence of a carbapenem-resistant Escherichia coli isolate with bla NDM-5
from a dog in the United States. Microbiol Resour Announc. 8,e00872–19.
10.1128/MRA.00872-19
Wan, S.Y., Hartmann,
F.A., Jooss, M.K. and Viviano, K.R. (2014). Prevalence and clinical outcome of
subclinical bacteriuria in female dogs. J. Am. Vet. Med. Assoc., 245, 106-112.
Way, L.I., Sullivan,
L.A., Johnson, V. and Morley, P.S. (2013). Comparison of routine urinalysis and
urine Gram stain for detection of bacteriuria in dogs. J. Vet. Emerg. Crit. Care, 23, 23–28.
Weese, J. S., Stull,
J. W., Evason, M., Webb, J., Ballance, D., McKee, T. and Bergman, P. J. (2022).
A multicenter study of antimicrobial prescriptions for cats diagnosed with
bacterial urinary tract disease. Journal of Feline Medicine and Surgery, 24(8),
806–814. doi.org/10.1177/1098612X211054815
Weese, J.S.,
Blondeau, J., Boothe, D., Breitschwerdt, E., Guardabassi, L., Hillier, A., Lloyd, D., Papich, M.G., Rankin,
S. and Turnidge, J.D. (2011). Antimicrobial use guidelines for treatment of
urinary tract infections in dogs and cats: antimicrobial guidlines working
group of the International Society for Companion Animal Infectious Diseases. Vet.
Med. Int., 4, 1–9.
Weese, J.S., Blondeau, J., Boothe, D.,
Guardabassi, L.G., Gumley, N., Papich, M., Jessen, L.R., Lappin, M., Rankin, S.
and Westropp, J.L. (2019). International
Society for Companion Animal Infectious Diseases (ISCAID) guidelines for the
diagnosis and management of bacterial urinary tract infections in dogs and
cats. Vet. J., 247, 8–25.
Werner, A. and
Norton, F. (2011). Blastomycosis. Compend Contin. Educ. Vet., 33(8),
E1-4.
White, J.D., Cave,
N.J., Grinberg, A., Thomas, D.G. and
Heuer, C. (2016). Subclinical bacteriuria in older cats and its
association with survival. J. Vet. Intern. Med., 30, 1824-1829.
Wong, C., Epstein,
S.E. and Westropp, J.L. (2015). Antimicrobial susceptibility patterns in
urinary tract infections in dogs (2010–2013). J. Vet. Intern. Med., 29,
1045–1054.
Wu, R., Dong, Z.,
Liu, Y., Xin, J., Duan, Y., Zheng, H., Yang, Y., Fu, H., Zhong, Z., Liu, H.,
Zhou, Z., Huang, Y. and Peng, G. (2024). Bacteriophage P2-71: a promising
therapeutic against multidrug-resistant Proteus mirabilis in urinary
tract infections. Frontiers in Veterinary Science, 11, 1445264.
doi.org/10.3389/fvets.2024.1445264
Yu, Z., Wang, Y.,
Chen, Y., Huang, M., Wang, Y., Shen, Z., Xia, Z. and Li, G. (2020).
Antimicrobial resistance of bacterial pathogens isolated from canine urinary
tract infections. Veterinary Microbiology, 241, 108540.
doi.org/10.1016/j.vetmic.2019.108540
Yudhanto, S., Hung,
C.C., Maddox, C.W. and Varga, C. (2022). Antimicrobial Resistance in Bacteria
Isolated From Canine Urine Samples Submitted to a Veterinary Diagnostic
Laboratory, Illinois, United States. Front Vet. Sci., 9,867784. doi:
10.3389/fvets.2022.867784.
Zaidi, S., Bouam, A.,
Bessas, A., Hezil, D., Ghaoui, H., Ait-Oudhia, K., Drancourt, M. and Bitam, I.
(2018). Urinary shedding of pathogenic Leptospira in stray dogs and cats,
Algiers: A prospective study. PLoS One,13,(5),e0197068. doi:
10.1371/journal.pone.01970.