Educational
Notes
Innate Immunity: Physical and Mechanical
Barriers
Abouelhag H. A*
Microbiology and Immunology
Dept., National Research Centre, Dokki, Giza, Egypt, 12622.
Received: 08-04-2026 Accepted: 22-04-2026 Published online: 30-04-2026
DOI: https://doi.org/10.33687/ricosbiol.04.04.121
Abstract
Innate immunity constitutes
the first line of host defense against invading pathogens (Turvey & Broide,
2010). Among its components, physical and mechanical barriers play a fundamental
role by preventing microbial entry, colonization, and dissemination. This educational
note provides a comprehensive overview of these barriers across different anatomical
sites, including the skin, mucous membranes, respiratory tract, gastrointestinal
tract, genitourinary tract, and eyes. It also details associated chemical factors
(e.g., lysozyme, lactoferrin, gastric juice, bacteriocins) and cellular elements
such as Langerhans cells, M cells, and alveolar macrophages (Abbas et al., 2020;
Gallo & Hooper, 2012). The note emphasizes the synergistic action of physical,
mechanical, and chemical mechanisms that together form an effective surveillance
system. Understanding these barriers is essential for appreciating how the body
resists infection before adaptive immunity is engaged.
Keywords:
: Innate immunity, physical barriers, mechanical barriers, skin, mucous
membranes, lysozyme, lactoferrin, M cells, alveolar macrophages, SALT, mucosal-associated
lymphoid tissue, antimicrobial peptides
Introduction to Innate Immunity
Innate immunity is the
evolutionarily ancient, non-specific defense system that responds immediately to
pathogens. Unlike adaptive immunity, it does not require prior exposure and lacks
immunological memory (Turvey & Broide, 2010). The physical and mechanical barriers
are the most external components of innate immunity, designed to prevent pathogen
entry or rapidly remove them before infection can establish (Abbas et
al., 2020).
These barriers include:
·
Intact skin and mucous
membranes.
·
Mechanical actions such
as shedding, flushing, ciliary movement, peristalsis, coughing, and sneezing.
·
Chemical factors that directly
kill or inhibit microbes (Gallo & Hooper, 2012).
Together, they provide
a formidable first line of defense.
1. The Skin: A Multilayered Physical and Chemical Fortress
The skin is the largest
organ of the body (≈1.5–2 m²) and serves as a primary physical barrier (Nestle et
al., 2009).
1.1 Structural Features
·
Keratinocytes form multiple layers of
stratified squamous epithelium; the outer layer (stratum corneum) is composed of
dead, keratin-filled cells that are impermeable to most microorganisms (Nestle et
al., 2009).
·
Continuous shedding of outer epithelial cells
removes attached microbes (Abbas et al., 2020).
·
Relative dryness (low water activity) slows
microbial growth (Gallo & Hooper, 2012).
·
Mild acidity (pH 5–6) – due to lactic
acid, free fatty acids, and amino acids – inhibits many pathogenic bacteria and
fungi (Schroder, 2011).
·
Sebum (from sebaceous glands)
contains triglycerides that are broken down into free fatty acids with antimicrobial
activity (Schroder, 2011).
·
Normal skin microbiota (e.g., Staphylococcus
epidermidis) produces bacteriocins and competes for nutrients, antagonizing
pathogens like Staphylococcus aureus (Gallo & Hooper, 2012).
·
Hygiene (washing) mechanically
removes transient microorganisms.
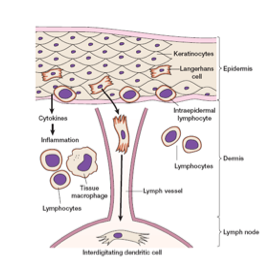
1.2 Skin-Associated Lymphoid
Tissue (SALT)
The skin is not just a
passive barrier; it contains specialized immune cells (Nestle et al., 2009):
·
Langerhans cells – dendritic cells in the
epidermis that phagocytose antigens, migrate to draining lymph nodes, mature into
interdigitating dendritic cells, and present antigens to naïve T cells, initiating
adaptive immunity (Abbas et al., 2020).
·
Intraepidermal lymphocytes – primarily γδ T cells
that act like cytotoxic T lymphocytes, destroying infected keratinocytes (Nestle
et al., 2009).
·
Large numbers of macrophages in the dermis that phagocytose
pathogens and produce inflammatory cytokines (Abbas et al., 2020).
2. Mucous Membranes and Mucosal-Associated Lymphoid Tissue (MALT)
Mucous membranes line internal
cavities exposed to the external environment (oral cavity, nasal passages, gut,
vagina, etc.). They are more delicate than skin but have specialized defenses (Mestecky
et al., 2015).
2.1 General Features
·
Mucus – a viscous secretion
containing glycoproteins (mucins) that traps microorganisms (Mestecky et al., 2015).
·
Antimicrobial components:
o
Cervical mucus – impedes ascent of bacteria
into the uterus.
o
Prostatic fluid – contains zinc and antibacterial
factors.
o
Tears – contain lysozyme, lactoferrin,
and sIgA (Kolar & McDermott, 2019).
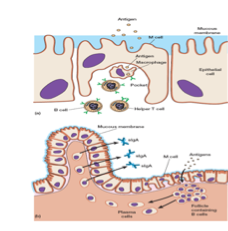
2.2 M Cells (Microfold Cells)
M cells are specialized
epithelial cells found overlying lymphoid follicles in the gut, tonsils, and Peyer’s
patches (Mestecky et al., 2015).
·
Structure: Lack microvilli (brush
border) but have a pocket on their basolateral side containing B cells, T cells,
and macrophages (Abbas et al., 2020).
·
Function:
1.
Phagocytose antigens and
pathogens from the gut lumen.
2.
Transport them across the
epithelial barrier into the pocket.
3.
Macrophages in the pocket
engulf the antigen.
4.
Alternatively, M cells
deliver antigens to organized lymphoid follicles.
5.
B cells in the follicle
recognize the antigen, mature into plasma cells, and secrete secretory IgA (sIgA)
(Mestecky et al., 2015).
6.
sIgA is transported into
the gut lumen to neutralize specific pathogens.
This mechanism is a critical
bridge between innate and adaptive immunity at mucosal surfaces (Abbas et al., 2020).
3. Defenses of the Respiratory System
The respiratory tract is
constantly exposed to airborne pathogens and particulates. Multiple barriers protect
it (Sarkar & Tindle, 2021).
3.1 Upper Respiratory Tract
·
Nasal cilia beat toward the pharynx,
moving mucus (with trapped microbes) to the mouth for swallowing or expulsion (Abbas
et al., 2020).
·
Humidification of inhaled air causes
hygroscopic (water-absorbing) microorganisms to swell, which may disrupt their membranes
and also facilitates phagocytosis.
·
Mucociliary blanket – a layer of mucus on
ciliated epithelial cells that traps particles <10 µm in diameter (including
most bacteria and viruses). Cilia beat at ≈1000 beats/min to propel mucus upward
(Sarkar & Tindle, 2021).
3.2 Reflexes
·
Coughing and sneezing reflexively
expel large amounts of air and mucus, removing irritants and pathogens (Abbas et
al., 2020).
3.3 Saliva
Saliva from the mouth washes
microorganisms from the oral and nasopharyngeal areas into the stomach, where gastric
acid destroys them (Gallo & Hooper, 2012).
3.4 Lower Respiratory Tract
and Alveolar Macrophages
· Alveolar macrophages reside in the lung alveoli. They are highly phagocytic
and can ingest and kill most bacteria via reactive oxygen species and lysosomal
enzymes. They also clear apoptotic cells and debris (Sarkar & Tindle, 2021).
· In addition, surfactant proteins (SP-A and SP-D) act as opsonins
that enhance phagocytosis (Abbas et al., 2020).
4. Gastrointestinal Tract Defenses
The GI tract faces a high
microbial load from ingested food and water. Its defenses are both physical and
chemical (Gallo & Hooper, 2012).
4.1 Chemical Barriers
·
Gastric juice (pH 1.5–3.5) – destroys
most ingested bacteria, parasites, and viruses. Only a few pathogens (e.g., Helicobacter
pylori, Vibrio cholerae) survive (Abbas et al., 2020).
·
Pancreatic enzymes (trypsin, chymotrypsin)
– digest bacterial cell walls and proteins.
·
Bile – contains bile salts
that disrupt bacterial membranes (Gallo & Hooper, 2012).
4.2 Mechanical Defenses
·
Peristaltic movement – propels intestinal contents
forward, preventing stasis and colonization (Abbas et al., 2020).
·
Intestinal microbiota (commensal bacteria) –
provide colonization resistance via:
o
Production of bacteriocins
(e.g., colicin from E. coli, staphylococcin from Staphylococcus) (Gallo
& Hooper, 2012).
o
Competition for nutrients
and adhesion sites.
o Stimulation of host immune responses (Mestecky et al., 2015).
5. Genitourinary Tract Defenses
The urinary and reproductive
tracts are protected by several features (Abbas et al., 2020):
·
Urine properties: Low pH (≈5.5–6.5), high
urea concentration, uric acid, and hippuric acid – all inhibit microbial growth.
·
Hypotonic effect of the kidney medulla
– creates osmotic stress for bacteria.
·
Flushing action – frequent voiding of
urine mechanically removes pathogens.
·
Distance barrier – long urethra (≈20 cm
in males) makes ascending infection more difficult.
·
Secretory antibodies (sIgA) in cervical mucus neutralize
sperm-borne and sexually transmitted pathogens (Mestecky et al., 2015).
·
Prostatic antibacterial
factor – a zinc-containing peptide with antimicrobial activity.
6. The Eyes: Continuous Cleansing
The ocular surface is constantly
exposed but remains remarkably infection-free due to (Kolar & McDermott, 2019):
·
Continuous flushing by tears (produced by
lacrimal glands, drained via nasolacrimal duct).
·
Tear composition:
o Lysozyme (muramidase) – breaks the β(1→4) bond between
N-acetylmuramic acid and N-acetylglucosamine in peptidoglycan, especially effective
against Gram-positive bacteria (Kolar & McDermott, 2019).
o Lactoferrin – iron-binding protein that sequesters iron, limiting
bacterial growth (Kolar & McDermott, 2019).
o sIgA – neutralizes pathogens and prevents adhesion
(Mestecky et al., 2015).
o Lactoperoxidase – generates superoxide radicals that kill microbes.
Thus, tears provide both
physical (flushing) and chemical protection.
7. Chemical Barriers: A Closer Look
While many chemical factors
are associated with specific sites, some are systemic or widely distributed (Gallo
& Hooper, 2012; Schroder, 2011).
|
Chemical Barrier |
Source |
Mechanism of Action |
|
Lysozyme |
Tears, saliva, mucus, milk |
Hydrolyzes peptidoglycan (Gram-positive bacteria) (Kolar &
McDermott, 2019) |
|
Lactoferrin |
Neutrophils, macrophages, secretions |
Iron chelation; disrupts bacterial membranes (Kolar & McDermott,
2019) |
|
Lactoperoxidase |
Saliva, milk, tears |
Generates hypothiocyanite and superoxide radicals |
|
Gastric juice |
Stomach |
Acid denaturation of proteins (Abbas et al., 2020) |
|
Salivary glycoproteins |
Saliva |
Inhibit bacterial adhesion |
|
Urea |
Urine |
Alkaline degradation products are antimicrobial |
|
Bacteriocins (colicin, staphylococcin) |
Commensal bacteria |
Pore formation, cell wall synthesis inhibition (Gallo &
Hooper, 2012) |
|
β-Lysin |
Blood platelets |
Disrupts microbial plasma membrane |
|
Leukins |
Neutrophils |
Cationic antimicrobial peptides (Schroder, 2011) |
|
Phagocytin |
Phagocytes |
Antimicrobial protein |
|
Prostatic antibacterial factor |
Prostate fluid |
Zinc-dependent antimicrobial activity |
8. Summary and Clinical Relevance
·
Physical and mechanical
barriers are the first and most immediate components of innate immunity (Turvey
& Broide, 2010).
·
They act by blocking
entry (skin, mucous membranes), removing microbes (shedding, cilia, flushing,
peristalsis), and killing (chemicals, phagocytes) (Abbas et al., 2020).
·
These barriers are not
passive; they include specialized immune cells (Langerhans cells, M cells, alveolar
macrophages) that initiate adaptive responses when breached (Nestle et al., 2009;
Sarkar & Tindle, 2021).
·
Defects in these barriers predispose to infection:
e.g., burns (loss of skin barrier), cystic fibrosis (impaired mucociliary clearance),
gastric acid suppression (increased risk of GI infections) (Gallo & Hooper,
2012).
Understanding these mechanisms
is crucial for developing strategies to prevent infections and for appreciating
how the body maintains homeostasis with the microbial world.
References
Abbas, A. K., Lichtman,
A. H., & Pillai, S. (2020). Cellular and molecular immunology (10th ed.).
Elsevier. [ISBN: 978-0323524577]
Gallo, R. L., & Hooper, L. V. (2012). Epithelial
antimicrobial defence of the skin and intestine. Nature Reviews Immunology,
12(7), 503–516. https://doi.org/10.1038/nri3228
Kolar, S. S., & McDermott,
A. M. (2019). Role of lactoferrin in the eye. Biometals, 32(3), 365–374.
https://doi.org/10.1007/s10534-019-00193-1
Mestecky, J., Strober,
W., Russell, M. W., Kelsall, B. L., Cheroutre, H., & Lambrecht, B. N. (Eds.).
(2015). Mucosal immunology (4th ed.). Academic Press. https://doi.org/10.1016/C2012-0-00539-1
Nestle, F. O., Di Meglio,
P., Qin, J. Z., & Nickoloff, B. J. (2009). Skin immune sentinels in health and
disease. Nature Reviews Immunology, 9(10), 679–691. https://doi.org/10.1038/nri2622
Sarkar, A., & Tindle,
C. (2021). Alveolar macrophages in lung inflammation and resolution. Frontiers
in Immunology, 12, 758789. https://doi.org/10.3389/fimmu.2021.758789
Schroder, J. M. (2011).
Antimicrobial peptides in the skin. Seminars in Immunopathology, 33(1), 3–13.
https://doi.org/10.1007/s00281-010-0210-5
Turvey, S. E., & Broide,
D. H. (2010). Innate immunity. Journal of Allergy and Clinical Immunology,
125(2 Suppl 2), S24–S32. https://doi.org/10.1016/j.jaci.2009.07.016
Data
Availability Statement
No
original datasets were generated for this review article. All cited data and findings
are available within the original research publications referenced in the manuscript,
accessible via the provided Digital Object Identifiers (DOIs) or through respective
journal platforms.